Philadelphia has not been traditionally known as a leading hub for biotechnology innovation and commercialization, in contrast to those hubs located Boston and San Francisco. However, the city is hoping that the more than 16,000 delegates attending the BIO 2019 International Convention this week will learn just how much research and development is taking place and that global recognition for its foundational research in gene and cell therapy has earned it the name of “Cellicon Valley.”
Certainly, the number of biotechnology companies located in the greater Philadelphia region has grown rapidly during the past few years and, according to the CEO Council for Growth, a council of the Chamber of Commerce for Greater Philadelphia, the region is now home to more than 30 cell and gene therapy companies.
 Jim Greenwood, president and CEO of the Biotechnology Innovation Organization (BIO), told BioWorld Insight that one of the interesting things about this year’s meeting is that attendees will gain an appreciation of the tremendous resurgence of gene therapy that has taken place in the area. That has evolved after a very difficult start almost 20 years ago when a young patient died as a result of the experimental therapy he received at the University of Pennsylvania.
Jim Greenwood, president and CEO of the Biotechnology Innovation Organization (BIO), told BioWorld Insight that one of the interesting things about this year’s meeting is that attendees will gain an appreciation of the tremendous resurgence of gene therapy that has taken place in the area. That has evolved after a very difficult start almost 20 years ago when a young patient died as a result of the experimental therapy he received at the University of Pennsylvania.
The 1999 death of 18-year-old Jesse Gelsinger came four days after researchers injected genes to cure ornithine transcarbamylase deficiency, a genetic metabolic disease. (See BioWorld Today, Dec. 10, 1999.)
The technology remained in the doldrums after that serious setback but slowly it has got off the floor and flourished thanks to breakthrough discoveries in cell and gene therapies, much of which originated in the region. That seminal work has culminated in several new medicines reaching the market, including Philadelphia-based Spark Therapeutics Inc.’s FDA approval for Luxturna (voretigene neparvovec-rzyl), which became the first gene therapy approved in the U.S. that targets an inherited disease caused by mutations in a specific gene. The treatment was approved to treat children and adults with confirmed biallelic RPE65 mutation-associated retinal dystrophy, an ultra-rare progressive disease that leads to vision loss and may cause blindness. (See BioWorld, Dec. 20, 2017.)
Unique period
Along with those developments, the pace of discovery continues in all areas of biotechnology. It is a unique period right now for the industry, Greenwood reflected, where the science has never been better but, unfortunately, at the same time, the prevailing political climate has never been more challenging as the debate on the current state of drug pricing rages on.
Those headwinds are likely to keep battering the industry and intensify as the upcoming 2020 U.S. presidential election campaign gets into full swing.
BIO 2018 – BIO 2019
Although Wall Street has acknowledged the political risk that swirls around the industry, it believes, for the time being, that something radical is not about to happen. However, “it won’t take much, in terms of bad policy related to health care, to spook investors,” Greenwood said.
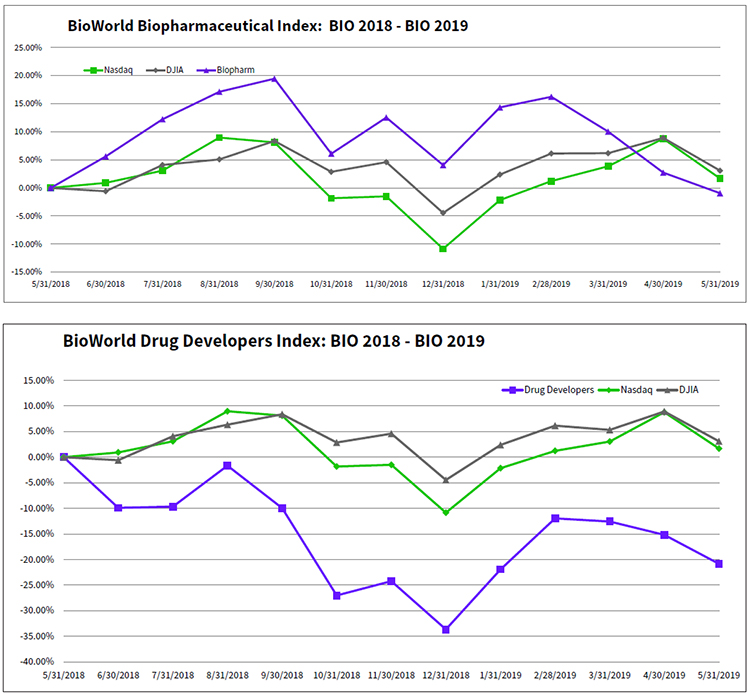
Since BIO 2018 closed it doors in Boston last June, public biopharmaceutical companies have experienced some good times as well as low periods, particularly in the fourth quarter of 2018 when investors shied away from biopharma company equities big time. As a result, the BioWorld Biopharmaceutical Index dropped almost 7% in December. The month will also go down in history as one of the worst periods for equities, with the Dow Jones Industrial Average stumbling by almost 9% and the Nasdaq Composite performing just as poorly, dropping 9.5%. (See BioWorld Insight, Jan. 7, 2019.)
Fortunately, the early months of this year saw investors return to the sector, anticipating that there will be an increase in the number of M&A transactions going forward. That belief was certainly reinforced with Roche Holding AG’s acquiring Spark Therapeutics, paying $114.50 per share in an all-cash deal valued at about $4.8 billion. Spark will operate as an independent firm inside Roche after the deal is completed. (See BioWorld, Feb 26, 2019.)
Over the past 12 months, the positive and negative swings in capital market performance have balanced out, with the BioWorld Biopharmaceutical Index closing the period down just 1% compared to the Dow Jones Industrial Average and the Nasdaq Composite index dipping 3% and 1.7%, respectively. (See BioWorld Biopharmaceutical Index BIO 2018 – BIO 2019, below.)
Turbulent markets hit drug developers
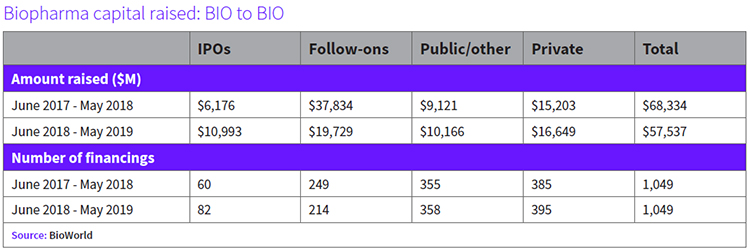
While there has been a resurgence of investor interest this year in small and midcap drug developers, the turbulent markets have hit those biopharmaceutical companies. “Generalist investors fear the constant scrutiny of the industry’s pricing practices and negative headlines as the Presidential primaries approach and so are looking for ideas in less-controversial sectors. They have not participated in any meaningful way in biotech year-to-date in 2019, and appear unlikely to return any time soon,” the Cowen and Co. biotechnology analyst team wrote in its April biotech thermometer report.
Overall, in the 12-month period since BIO 2018, the BioWorld Drug Developers Index recorded a 21% drop in value. (See BioWorld Drug Developers Index BIO 2018 – BIO 2019, below.)
Cash flows
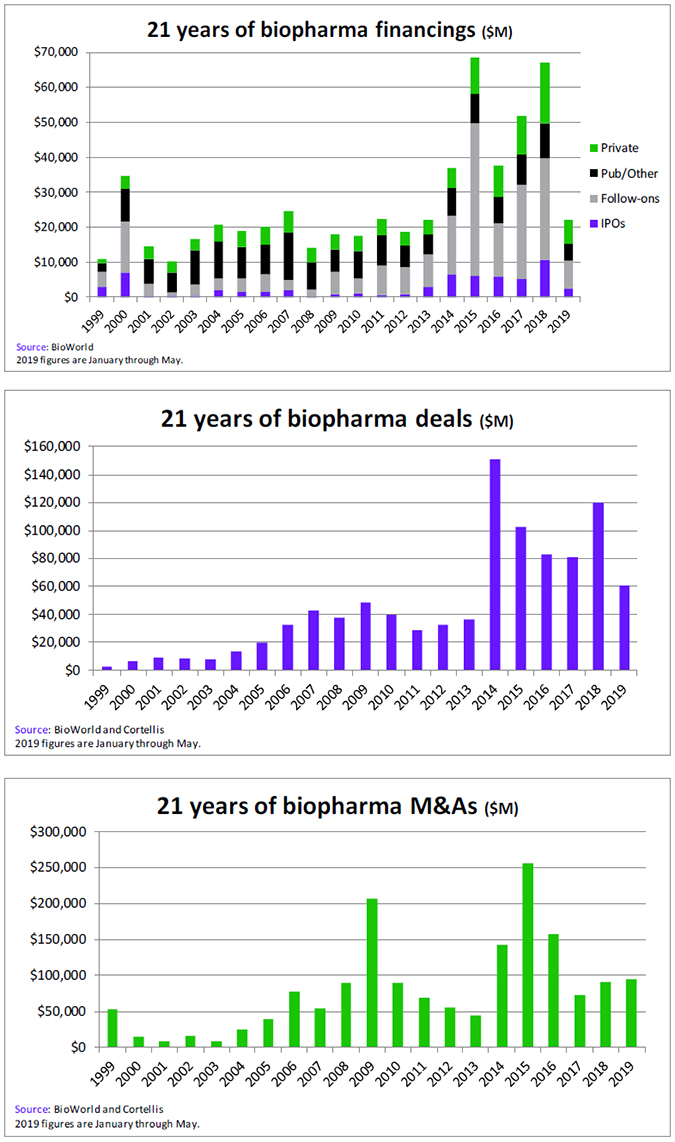
Despite the uncertainties of the capital markets during the past 12 months, public and private biopharma companies have had no difficulty raising capital – almost $58 billion, in fact, generated from 1,049 transactions, according to data from BioWorld. (See Biopharma capital raised: BIO to BIO, below. )
Of that amount, approximately 71% was raised by companies completing public offerings, including follow-on financings and IPOs. It is notable that the turbulence experienced in the general markets did not serve to dampen the enthusiasm for IPOs, with a steady flow of biopharma company IPOs completed throughout the year. A total of 82 companies closed their offerings, generating almost $11 billion in the process, compared to the almost $6.2 billion generated in the BIO 17-BIO 18 period.
Private companies have also captured a record amount of capital. The 395 companies that have raised cash since BIO 2018 have brought in a massive $16.6 billion. This year alone global private companies attracted more than $4.4 billion in the first quarter, 10% more than the $4 billion generated in the first quarter last year. In fact, it is also the highest amount raised in this period in the past decade. That total could signal that the sector is well on its way to surpass the record $17 billion private financings set in 2018.
By the numbers
Since the BIO International Convention was last held in Philadelphia in 2015, the industry has been on a record-setting pace for generating capital. That year, the high bar was set with a total of $68.4 billion raised. Last year, according to BioWorld data, $67.1 billion was raised by global companies from public and private sources, which places it firmly in second place in terms of totals raised in the history of the industry. (See 21 years of biopharma financings, below.)
The $20 billion raised this year to date means that in the past five years the global biopharma industry has generated an incredible $250 billion from its fundraising activities.
Business development has been equally robust in that period, with the value of partnering transactions averaging about $90 billion annually. (See 21 years of biopharma deals, below.)
The expectation by investors that mergers and acquisitions will pick up speed appear to have been confirmed, with the 2019 total of M&A transactions already outpacing the 2018 total. (See 21 years of M&As, below.)
Let’s make a deal
Greenwood noted that the annual convention’s One-on-One Partnering has evolved into an important component of the BIO meeting. At last year’s event, more than 46,000 meetings were recorded, which set the Guinness World Record for the “Largest Business Partnering Event.”
The meeting in Philadelphia is already on pace to surpass that record, he said.
Those invaluable connections form the foundation of innovative industry collaborations and scientific breakthroughs and BIO has selected the theme of “It starts with one” for this year’s event. Greenwood explained that it only needs one molecule, one scientist, one conversation that can lead to the creation of one company that could create a lifesaving new medicine.