SAN FRANCISCO – One of the takeaway messages for delegates attending the recent Allicense 2015 meeting was that M&As remain on a tear right now and it is predicted that their numbers are likely to outpace last year's record transactions.
While it remains the case that significant M&A activity involves companies that have lead products in their pipeline at phase II and others that have products on the market, 2014 saw a marked shift to early stage M&A; that trend appears to be continuing into this year, noted Laura Vitez, principal business analyst, Thomson Reuters Recap, who dissected the current biopharma deal landscape referencing Recap's first quarter 2015 analysis.
Adding to the trend toward early stage assets was Bristol-Myers Squibb Co. (BMS), which shelled out $800 million up front and as much as $450 million more earmarked for milestone payments, for privately held Flexus Biosciences Inc., of San Carlos, Calif. The deal gives BMS full rights to the firm's indoleamine 2,3-dioxygenase 1 (IDO1)/tryptophan 2,3-dioxygenase (TDO) discovery program, which includes IDO-selective, IDO/TDO dual and TDO-selective compound libraries. From the platform, the lead preclinical small-molecule IDO1, F001287, is expected to become the subject of an investigational new drug application (IND) in the second half of this year. (See BioWorld Today, Feb. 24, 2015.)
Overall, the data showed that of the 891 deals in the period captured by Recap from the entire life sciences industry, including therapeutics, medical devices, diagnostics and contract research organizations, approximately 13 percent of the deal volume comprised M&As. Interestingly, thanks to several blockbuster transactions, those deals represented more than 72 percent of the $99.3 billion total deal value in the quarter.
RARE DISEASES
Putting an exclamation point on the M&A year-to-date total was Alexion Pharmaceutical Inc.'s announcement of an $8.4 billion cash-and-stock buyout of Lexington, Mass.-based Synageva Biopharma Corp., consisting of a premium $115 in cash plus 0.6581 share for each share of Synageva. The price equated to a share value of $230 based on the nine-day average closing price of Alexion. (See BioWorld Today, May 7, 2015.)
 Both companies operate in the rare diseases space, which has been a hotbed, leading to more than $18 billion in combined acquisition deal value so far this year. (See Leading Biopharma M&A Deals table, right.)
Both companies operate in the rare diseases space, which has been a hotbed, leading to more than $18 billion in combined acquisition deal value so far this year. (See Leading Biopharma M&A Deals table, right.)
Synageva adds Kanuma (sebelipase alfa) for lysosomal acid lipase deficiency (LAL-D) to Alexion's metabolic franchise. The product is expected to hit the market later this year. That's also when Alexion's Strensiq (asfotase alfa) for hypophosphatasia should reach the market. In the combined pipeline are eight product candidates in the clinic for 11 indications.
Kanuma is under priority review with the FDA and has won accelerated consideration of its marketing authorization application by the EMA. U.S. regulators also have designated the drug a breakthrough therapy for LAL-D in infants. Regulatory decisions in the U.S. and Europe are expected in the second half of the year.
Shire plc kicked off the quest for rare disease-focused companies at the beginning of 2015 with its takeover of NPS Pharmaceuticals Inc. for $5.2 billion in cash, gaining access to NPS' portfolio of treatments for rare gastrointestinal (GI) disorders.
NPS's lead product, known as Gattex in the U.S. and Revestive in Europe (teduglutide of rDNA origin), is approved for the treatment of short bowel syndrome for patients who are dependent on parenteral support. Shortly after the deal was announced the company received FDA approval of Natpara, a recombinant human parathyroid hormone (PTH), as an adjunct to calcium and vitamin D to control hypocalcemia in patients with hypoparathyroidism. The rare endocrine disorder is characterized by insufficient levels of PTH. (See BioWorld Today, Jan. 27, 2015.)
Rounding out the first quarter was Jerusalem-based pharma giant Teva Pharmaceutical Industries Ltd.'s $3.2 billion acquisition of orphan drug firm Auspex Pharmaceuticals Inc. In the merger, Teva brings aboard La Jolla, Calif.-based Auspex's lead compound, SD-809 (deutetrabenazine), which grew out of the company's deuterium technology platform and is being developed for the potential treatment of chorea associated with Huntington's disease, tardive dyskinesia and Tourette syndrome, with a pharmacokinetic profile that allows for lower doses and thus potential for an improved safety profile.
Given the prevailing M&A environment and continuing enthusiasm for IPOs, albeit at a slower pace than last year, executives of emerging biotech companies have the luxury of being able to opt for an IPO or consider overtures for an acquisition.
"There is certainly a lot of early stage exit activity out there and, based on current transactions, M&A activity is likely to outpace 2014," said Jonathan Norris, managing director of the health care practice at Silicon Valley Bank, in a presentation on venture financing trends and M&A.
SLOW START FOR IPOs
While companies with quality early stage assets are potential takeout targets, the IPO route is still available for them despite the slow start for those offerings in the wake of choppy financial markets.
Based on the first quarter transactions, Norris projected there could be at least 40 IPOs by year end.
In fact, we are already at nearing halfway point in that prediction thanks to a flurry of activity last week.
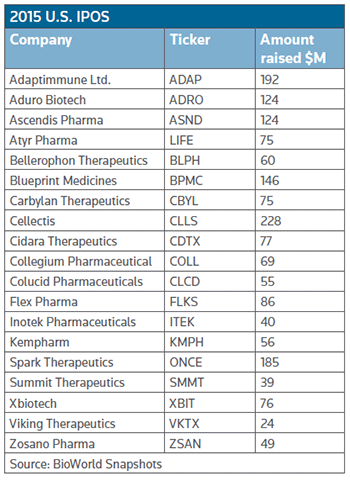 According to BioWorld Insight analysis, there have been 19 companies who have made the transition to the U.S. public arena so far, compared to 34 that had completed their IPOs at the same point last year. (See 2015 U.S. IPOs, right.)
According to BioWorld Insight analysis, there have been 19 companies who have made the transition to the U.S. public arena so far, compared to 34 that had completed their IPOs at the same point last year. (See 2015 U.S. IPOs, right.)
At the close of last week, Atyr Pharma Inc. and Collegium Pharmaceutical Inc. got their deals done, raising similar amounts of capital, although neither was able to push pricings to the top of their range in a market that has become more cautious to biotech offerings of late. (See BioWorld Today, May 7, 2015.)
Atyr priced 5.36 million shares at $14 each to raise just over $75 million, while Collegium collected almost $70 million by pricing 5.8 million shares at $12 each. A day earlier Colucid Pharmaceuticals Inc. and Adaptimmune Ltd. raised $55 million and $192 million, respectively.
San Diego-based Atyr, which is now trading on Nasdaq under the ticker LIFE, is developing Resolaris, which has orphan drug designation from the FDA in facioscapulohumeral muscular dystrophy (FSHD), the focus of an ongoing phase Ib/II trial.
The company granted the underwriters a 30-day option to purchase up to 804,000 additional shares as overallotments, potentially adding approximately $11.3 million in additional funds from the IPO.
Collegium's pipeline is focused on candidates that incorporate its Deterx technology platform, using the 505(b)(2) pathway. The oral drug delivery technology is designed to provide extended-release delivery, abuse-deterrent properties and flexible dose administration options to treat chronic pain, with its formulation of oxycodone now under review at the FDA. The Canton, Mass.-based company granted underwriters a 30-day option to purchase up to 870,000 additional common shares, potentially adding $10.44 million to the IPO raise.
Burlington, Mass.-based Colucid had to make some last-minute adjustments, lowering its price to $10 from its hoped-for range of $13 to $15 and raising the number of shares offered from 5.4 million to 5.5 million. It also got a little help from insiders, with existing shareholders agreeing to purchase up to $25 million – about 45.5 percent – of the $55 million offering.
Adaptimmune, meanwhile, priced at the high end of its proposed range, selling 11.3 million American depositary shares – upsized from 9.4 million – at $17 each for gross proceeds of $192 million. The Abingdon, UK-based firm is working in the cancer immunotherapy space, which remains a favorite technology with investors.
ENCOURAGING NEWS
The 2015 graduating U.S. public companies have collectively raised $1.6 billion at an average of $83.5 million per transaction.
Investors will be encouraged to keep supporting biotech IPOs going forward given the fact that the group's average shows an 11.2 percent increase in share values. In fact, only seven newly minted public companies were trading below their issue price at market close Thursday.
Top gainer is Philadelphia-based Spark Therapeutics Inc. whose shares (NASDAQ:ONCE) were trading up a whopping 153 percent from their $23 initial offering price, despite a few fleeting jitters last week stemming from disappointing data from an unrelated gene therapy. (See BioWorld Today, May 5, 2015.)
Spark's lead candidate, SPK-RPE65, is in a pivotal phase III program and focused on a group of rare blinding conditions.
The hot immune-oncology space also has helped vault Aduro Biotech Inc.'s (NASDAQ:ADRO) shares since its public debut. Its shares have gained 68 percent. With plenty of news ready to emerge from abstracts due to be released for the upcoming American Society of Clinical Oncology meeting it is likely that the biotech IPO momentum will not wane anytime soon.
