A flurry of FDA drug approvals the week leading up to the holiday break helped boost the total of new molecular entities that rolled out in 2014. The most recent was the Dec. 24 approval of Namzaric, a once-daily fixed-dose combo of Namenda XR and donepezil, for Alzheimer's disease dementia, brought good cheer to Adamas Pharmaceuticals Inc., of Emeryville, Calif. Partnered with Actavis plc (through the Dublin-based firm's buyout of Forest Laboratories Inc.), Namzaric, which combines the NMDA receptor antagonist memantine hydrochloride extended-release drug marketed by Actavis as Namenda with acetylcholinesterase inhibitor donepezil hydrochloride, was cleared for use in moderate-to-severe dementia of the Alzheimer's type in patients stabilized on memantine hydrochloride and donepezil hydrochloride.
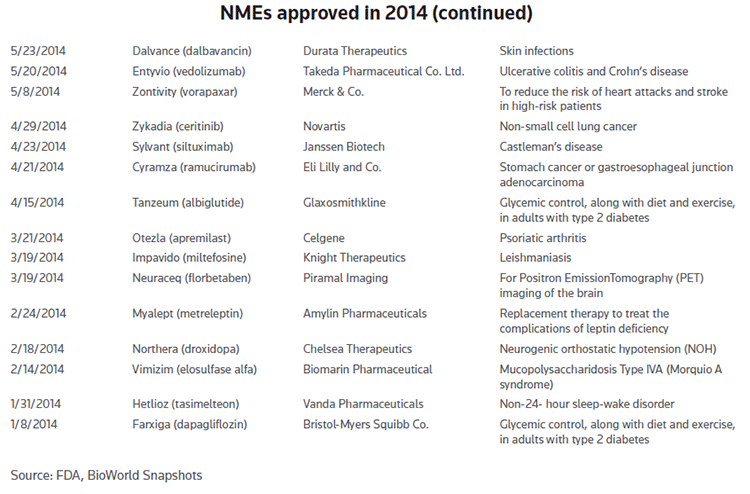
With just three days left this year the agency has now already given the green light to no less than 42 new molecular entities (NMEs) representing a 52 percent increase over the total number of NMEs that were approved last year. (See NMEs approved in 2014.)
ANTIBIOTICS
With worldwide concern about the growing number of infections that are now increasingly resistant to our current inventory of antibiotics, there will be comforting news in the fact that several new antibacterial drugs received approval this year.
Last week Cubist Pharmaceuticals Inc., of Lexington, Mass., received FDA approval of Zerbaxa (ceftolozane/tazobactam) for the treatment of adults with complicated urinary tract infections (cUTIs) and complicated intra-abdominal infections (cIAIs) caused by designated susceptible gram-negative bacteria. The FDA approval was supported by positive data from two pivotal phase III trials in patients with cUTI and cIAI, respectively. Both trials met the pre-specified primary endpoints agreed upon with the FDA and EMA. Zerbaxa, the company noted, is the first new antibiotic approved in the U.S. under the Generating Antibiotic Incentives Now (GAIN) Act to treat gram-negative bacteria and is designated by the FDA as a qualified infectious disease product for its indications. The drug became their second antibiotic to have received FDA approval this year. In June Cubist gained FDA approval for Sivextro (tedizolid phosphate), which provides the first big payoff from its $704 million acquisition of Trius Therapeutics Inc. (See BioWorld Today, June 24, 2014.)
The once-daily oxazolidinone antibiotic for treating certain gram-positive bacterial infections, including methicillin-resistant Staphylococcus aureus (MRSA), was one of the first antibiotics approved under the GAIN provision of the FDA Safety and Innovation Act.
Sivextro is approved to treat patients with acute bacterial skin and skin structure infections (ABSSSIs) caused by certain susceptible bacteria, including S. aureus, various Streptococcus species, and Enterococcus faecalis. Sivextro is available for intravenous and oral use.
A month earlier, the agency approved Durata Therapeutics Inc.'s Dalvance (dalbavancin), which represented the first qualified infectious disease product (QIDP) to win approval, getting the nod for use in ABSSSIs caused by gram-positive microorganisms, including the dreaded MRSA. (See BioWorld Today, May 27, 2014.)
Rounding out the new antibiotics medicine chest was The Medicines Co.'s antibiotic Orbactiv (oritavancin) that received approval to treat ABSSSIs outside hospitals.
The therapy is approved to treat ABSSSIs caused by gram-positive microorganisms, including MRSA. Its label includes a warning regarding interference with coagulation tests and interaction with warfarin, a drug used to prevent blood clots. (See BioWorld Today, Aug 7, 2014.)
Orbactiv was approved based on data from two trials, which showed that a single 1,200-mg dose of Orbactiv was non-inferior to twice-daily intravenous dosing of vancomycin given for seven to 10 days in patients with acute bacterial skin and skin structure infections caused or suspected to be caused by such gram-positive bacteria including MRSA.
PD-1 INHIBITORS
This year's crop of new medicines include drugs with blockbuster potential including Merck & Co. Inc.'s Keytruda (pembrolizumab) for treating melanoma. It is being hailed as a promising drug in its class – PD-1 inhibitors – which are designed to block the protein programmed death receptor-1, employed by tumors to avoid immune system defenses. Analysts are forecasting such inhibitors, which have potential as both monotherapies and part of combo therapies, as a multibillion-dollar market opportunity.
Merck also was successful with the approval of Zontivity (vorapaxar) tablets to reduce the risk of heart attack, stroke, cardiovascular death, and need for procedures to restore the blood flow to the heart in patients with a previous heart attack or blockages in the arteries to the legs.
A protease-activated receptor-1 (PAR-1) antagonist, a new class of drug, it is an anti-platelet agent, designed to decrease the tendency of platelets to clump together to form a blood clot.
Gilead Sciences Inc. scored big with two blockbuster potential drugs: Zydelig (idelalisib) its PI3k inhibitor for three types of B-cell blood cancers and Harvoni, the first oral therapy for people with genotype 1 hepatitis C virus (HCV) to eliminate the need for both interferon and ribavirin. The once-daily pill combines a dose of sofosbuvir, the active ingredient in Gilead's blockbuster HCV drug Sovaldi, with ledipasvir, a direct-acting macrocyclic antiviral agent and an inhibitor of NS5A serine protease.
HCV genotype 1 is the most common strain of the disease in developed nations, including the U.S., where an estimated 3.2 million people have chronic HCV infection, though many don't realize it until liver damage caused by the virus becomes apparent, which can take decades.
Based on Gilead's tests of Harvoni, the combination has the potential to cure HCV in as little as eight weeks, a feat that, when possible, would lower the overall cost of the treatment. Both drugs in Harvoni interfere with the enzymes needed by HCV to multiply. (See BioWorld Today, Oct. 13, 2014.)
PRODUCTIVITY UP FOR BIOTECHS
It hasn't been just big pharma companies stealing the limelight for getting new medicines "over the goal line." In fact 52 percent of the approvals this year were down to biotech companies. Gilead notched two as did Cubist Pharmaceuticals. Among the pharma companies, Merck scored with three approvals as did Boehringer Ingelheim GmbH.