A study led by Chinese radiologists at Peking University in Beijing has shown that positron emission tomography (PET) imaging of intercellular adhesion molecule-1 (ICAM-1) expression is a predictor for the abscopal effect, whereby nonirradiated cancers respond to radiotherapy (RT).
"To the best of our knowledge, this is the first study to report that PET of ICAM-1 expression allows early prediction of the abscopal effect of RT," said study leader Zhaofei Liu, a professor in the Department of Radiation Medicine of the School of Basic Medical Sciences at Peking University Health Science Center.
This finding may facilitate high-throughput screening of current or novel drugs for use in combination with RT, to achieve improved antitumor therapeutic outcomes that eliminate both primary and metastatic lesions, the authors reported in the April 6, 2021, edition of Proceedings of the National Academy of Sciences.
"The abscopal effect only occurs under rare circumstances in clinical practice, and the underlying mechanisms are neither fully understood nor therapeutically exploited," Liu told BioWorld Science. There are also no reliable biomarkers for early prediction of abscopal effects.
"Therefore, we need an effective means to predict the abscopal effect at an early RT stage, which would facilitate patient stratification and rational design of optimal combination regimens," he said.
"Unlike other ICAM-1 detection methods such as immunohistochemistry followed by invasive biopsies, which cannot accurately measure whole tumor ICAM-1 levels due to tumor heterogeneity, PET is noninvasive and can measure whole-body ICAM-1 dynamics."
Although RT has long been used as a frontline treatment for more than one-half of patients with malignant tumors, its efficacy is largely limited to localized tumors.
There is now evidence that, besides ablation of irradiated tumors, RT has a systemic abscopal inhibitory effect on nonirradiated lesions, the harnessing of which could systemically eliminate metastatic lesions disseminated beyond the radiation field.
In their new PNAS study, Liu and his team demonstrated that ICAM-1, an inducible glycoprotein member of the immunoglobulin superfamily, was upregulated in nonirradiated tumors responsive to RT.
"According to our proteomics results and those of ICAM-1-targeted near-infrared fluorescence (NIRF) imaging, ICAM-1 expression levels in nonirradiated tumors responsive to RT were approximately two-fold higher than in nonirradiated tumors unresponsive to RT," said Liu.
"The differences between the two groups can be sensitively detected by ICAM-1-targeted imaging, which can be used to predict the abscopal effect before tumor size divergence."
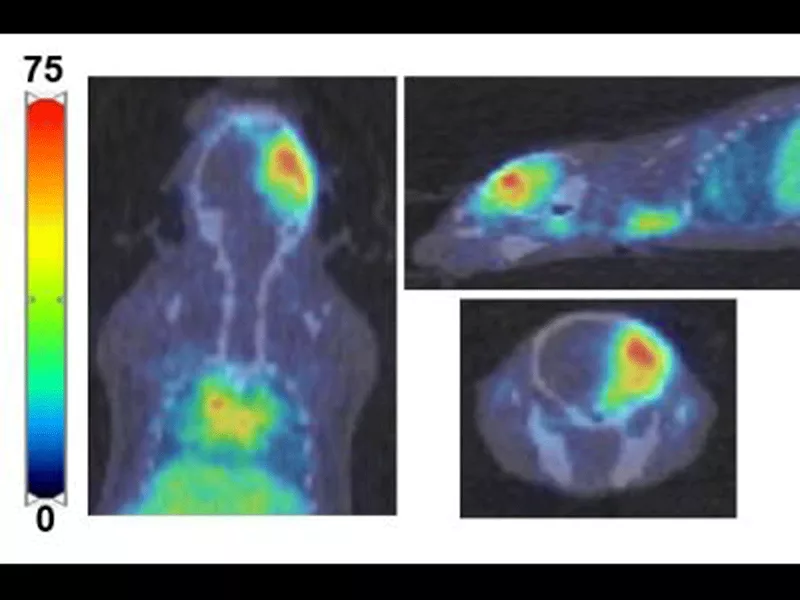
The researchers then showed that ICAM-1 expression could be noninvasively detected by optical imaging and PET in two preclinical animal models, using newly synthesized NIRF dye and 64Cu-labeled imaging probes.
I-CAM can
The authors tested their idea in two preclinical animal models: a 4T1 murine breast tumor model and a CT26 murine colon tumor model.
"To investigate whether noninvasive imaging could be used for in vivo ICAM-1 imaging, we synthesized the optical and PET imaging probes in this study," he explained.
"These probes were specific to murine ICAM-1 and can only be used to detect ICAM-1 in mouse models, so to facilitate clinical translation, we are now developing novel human ICAM-1-specific PET imaging probes in our lab."
Importantly, expression levels of ICAM-1 determined by quantitative PET imaging showed a strong negative linear correlation with growth of nonirradiated tumors.
"We found a negative correlation between tumor uptake of the ICAM-1-targeted probe on day 12 and the relative tumor volume changes 8 days later, suggesting the response of non-irradiated tumors to RT or combined RT therapy could be predicted early by ICAM-1 PET imaging," said Liu.
This is important, as "early prediction and precise monitoring of the abscopal effect of RT would facilitate patient stratification and help in designing optimal combination regimens," he said.
Moreover, genetic or pharmacologic upregulation of ICAM-1 expression by either an intratumoral injection of engineered recombinant adenovirus or systemic administration of a Toll-like receptor 7 agonist-capsulated nanodrug, were shown to induce markedly increased abscopal responses in local RT in the two animal models.
"These results show that ICAM-1 upregulation could augment he RT-induced abscopal effect, which suggests that ICAM-1 is a target to improve systemic antitumor effects of RT and may facilitate drug development," Liu said.
"For example, PET imaging of ICAM-1 expression levels may allow potential high-throughput screening of current or novel drugs to be used in combination with RT to achieve desired antitumor therapeutic outcomes that eliminate both primary and metastatic lesions."
Mechanistic studies revealed that ICAM-1 expression could enhance both the activation and tumor infiltration of CD8+ killer T cells to improve the responses of the non-irradiated tumors to RT.
Together, these findings suggest that noninvasive PET imaging of ICAM-1 expression could be a powerful means to predict the responses of non-irradiated tumors to RT, which could facilitate use of new combination RT strategies for effective ablation of primary and disseminated lesions.
"We have demonstrated that ICAM-1 upregulation is a potential biomarker for the RT-induced abscopal effect, predicting the response of metastatic lesions to RT by PET monitoring of ICAM-1 expression in the clinic," concluded Liu.
"ICAM-1 represents an attractive target for interventions to amplify the abscopal response of RT, which may guide the optimization of radiation doses, fractionation and combined therapy," he said.
"We are now developing new clinically applicable PET imaging probes targeting human ICAM-1, and we hope that PET of ICAM-1 expression using these can guide RT combination therapy in the clinic, to better serve cancer patients."