CDU Contributing Editor
ORLAND, Florida The 2005 scientific sessions of the American College of Cardiology (ACC, Bethesda, Maryland), held here in March, highlighted new developments in the rapidly growing areas of drug-eluting stents and implantable cardioverter defibrillators (ICDs), two of the most rapidly growing segments of the interventional cardiology market. Those segments, however, comprise only a part of the diverse market for cardiovascular devices, a market that is rapidly expanding in scope and in the range of applications addressed by suppliers. Other emerging growth segments in the cardiovascular device market include endovascular grafts, particularly new devices for treatment of thoracic aortic aneurysms; heart valves that can be implanted using percutaneous techniques; new circulatory support devices; embolic protection devices; thrombectomy catheters; devices for delivering cell-based therapy, and new cardiovascular surgery devices. Those segments provide significant opportunities for a number of suppliers in the market, including some new entrants.
New segments for cardiovascular market
Percutaneously implanted heart valves comprise one of the emerging segments of the cardiovascular device market attracting considerable investment focus. Phillipe Bonhoeffer, MD, of Emory University Hospital (Atlanta), described results of recent studies comparing percutaneous pulmonary valve implantation with surgical correction in 75 patients at the ACC sessions. A significant reduction in procedural complications was observed with the device. There was no mortality reported for the group treated with the percutaneous technique vs. 1.7% mortality for surgically treated patients, and a two-fold higher rate of other complications with surgery. The major drawback of the percutaneous valve is stenosis that occurs when a patient who is typically treated at a young age outgrows the valve. Some 91.7% of patients were free from device explant at one year in the study conducted with the device. Alain Cribier, MD, of Charles Nicolle Hospital (Rouen, France), described studies with a bioprosthetic aortic valve under development by Edwards Lifesciences (Irvine, California). The Cribier-Edwards aortic valve consists of an equine pericardium tricuspid valve attached to a stainless steel stent. The valve is implanted using a balloon catheter through a 22 Fr sheath. The life of the valve after implant is estimated at in excess of 10 years, according to Cribier.
Percutaneous repair of heart valves defects opens up a new segment of the market, allowing treatment of patients having a variety of contraindications for valve surgery such as age greater than 80 years, depressed left ventricular function, renal failure, cancer, respiratory failure, stroke, prior thoracotomy, and inability to pay for surgical treatment. The Edwards device, originally developed by Percutaneous Valve Technologies (Fort Lee, New Jersey), a company acquired by Edwards last year, can be implanted via either an antegrade or a retrograde approach. The heart is paced rapidly, at 220 beats per minute, to stop blood flow and allow accurate positioning of the device prior to deployment. Cribier implanted the Edwards/PVT valve in 32 patients between April 2002 and January 2005, with a mean patient age of 84 years and all diagnosed with NYHA Class IV heart failure. Three patients have survived in excess of one year post-implant. One factor cited by Cribier that will provide further improvement in outcomes is a more accurate pre-operative anatomic assessment of the native valve in order to ensure compatibility with the implanted device.
Another new percutaneous valve is under development by Advanced Bioprosthetic Surfaces Ltd. (ABPS; San Antonio, Texas). The PercValve is a nitinol device with leaflets that can be seeded with cells to create a tissue-engineered structure that can be implanted percutaneously. Good results were reported at the ACC sessions using the ABPS device. The company is using 3-D computer-aided design methods to optimize the fabrication of the valve leaflets. Cardiac Dimensions (Kirkland, Washington) is developing a percutaneous annuloplasty device that can be delivered using a 9 Fr catheter in patients with the proper heart anatomy. Pre-clinical studies with the device have allowed determination of the most appropriate types of patients for use of the annuloplasty device, and the company is proceeding with a clinical trial.
Mitralign (Salem, New Hampshire) is developing a percutaneous mitral valve for the treatment of mitral regurgitation. The Mitralign device is implanted via a transventricular retrograde approach, and three magnets are used to guide the placement catheter into the proper position for valve deployment. Tissue anchors are then placed to fix the valve in position. The company was formed about one year ago, and is currently performing animal studies with the mitral valve.
Ted Feldman, MD, of Evanston Hospital (Evanston, Illinois), discussed percutaneous techniques for performing edge-to-edge repair of the mitral valve using the EValve device. One application of such techniques, originally developed by Alfieri, is for correction of recurrent mitral regurgitation in patients who have undergone prior surgical repair. Recurrent regurgitation results in significant compromise of valve function in many patients who are initially treated with valve surgery. The device used by Feldman is an Elgiloy/polyester clip that is placed under transesophageal echocardiography guidance to coapt the mitral leaflets. It is being evaluated in the Endovascular Valve Edge-to-Edge Repair Study (EVEREST) trial, with 30-day results on the first 20 patients to receive implants presented at the ACC sessions. Clips have been placed in 17 of the 20 patients. Immediate reduction of mitral regurgitation was achieved in 76% of the patients, including two patients who received two clips. The mean length of hospital stay was 1.7 days.
Devices for the treatment of mitral regurgitation address a target patient population of 800,000 heart failure patients in the U.S. alone, as shown in Table 1. In most cases, the defect is not actually a valve abnormality, but rather is due to the enlargement of the myocardium that occurs in advanced heart failure. The only treatment option at present is open surgery, and 30% to 40% of patients who are treated surgically have recurrent regurgitation. In addition, patient survival is not improved by existing surgical treatments. Percutaneous technologies can potentially be used to treat recurrent regurgitation in patients who are no longer candidates for open surgery, as well as to treat patients with valve disease who are not surgical candidates or who desire a less invasive alternative.

Other percutaneous heart valve repair devices now in development include the Percutaneous Mitral Angioplasty device from Viacor (Wilmington, Massachusetts), the Entrata percutaneous device for the treatment of aortic valve disease under development by 3F Therapeutics (Lake Forest, California), and the PAVR ReValving System from CoreValve (Paris). The 3 Fr Entrata device is implanted through the apex of the aortic ventricle. Challenges include the need for resection of the native valve, fixation and positioning. However, the device could provide a revolution in treatment for aortic valve disease, according to presenters at the ACC conference.
First-in-man studies with the CoreValve device were reported at the ACC sessions by Eberhard Grube, MD, of Heart Center Siegburg (Siegburg, Germany). The ReValving system is comprised of a bovine pericardium valve mounted on a self-expanding frame in a 24 Fr catheter delivery system. The device was evaluated in a feasibility study in 21 patients with good results, including patients with both calcified aortic stenosis and aortic regurgitation. Good adhesion of the device to surrounding tissues was observed, and there was no device migration. Grube concluded that the CoreValve device may be a viable alternative to open-heart surgery. Technological advances are needed in areas such as 3-D echocardiography guidance and hemodynamic support to allow successful widespread use of such devices.
Another important emerging segment of the cardiovascular device market discussed at the ACC sessions is endovascular grafts for the treatment of thoracic aortic aneurysms (TAAs). The first TAA graft to be commercialized, the TAG Thoracic Endoprosthesis from W.L. Gore (Flagstaff, Arizona), was approved by the FDA in December 2004, marking a further expansion of the growing market for endovascular grafts. The Gore TAG consists of a nitinol stent with an ePTFE covering on the inner lumen. Although Gore has not yet begun shipment of the TAG to U.S. hospitals, cardiologists and related specialists anticipate rapid growth in implants once the device is widely available. Medtronic (Minneapolis), the current market leader in endovascular grafts, is also developing a device for TAA use, the Talent Thoracic Stent Graft System. Degenerative TAA has an incidence of 10.4% per 100,000 person-years. At present, patients who present with TAA, either due to degenerative disease or trauma, typically cannot be treated with an endovascular graft because implants can only be performed under the IDE protocol for the Gore device. Once the device is widely available, physicians who have used the TAG in clinical trials believe such patients will receive an immediate implant in order to avoid the risk of life-threatening rupture. The studies conducted by Gore that led to FDA approval of the TAG showed a highly statistically significant drop in major adverse events for patients receiving the graft vs. those treated surgically. Long-term, devices such as the Gore TAG are expected to replace open surgery for TAA repair, leading to a major increase in TAA repair procedures because incidence is under-reported at present. Table 2 presents estimates of recent trends in device sales in the endovascular graft market.

New devices for circulatory support also were highlighted at the ACC sessions. Orqis Medical (Lake Forest, California) exhibited the Cancion CRS, a new implantable pump for support of patients with acute heart failure. The device is intended for temporary (four-day) implantation, and has been evaluated as an alternative to medical therapy. The Cancion CRS uses two 12 Fr catheters, and provides continuous blood flow. It does not directly interface to the heart, but provides normal blood flow to other organs in the body, allowing the heart to rest and recover as it continues to function. Some 200,000 heart failure patients in the U.S. are non-responsive to medical management, making them potential candidates for use of the Orqis pump, and Medicare reimbursement is available for the device.
Another device for temporary circulatory support, the Recover LP25 from Impella/Accelerated Technologies (Salem, Massachusetts), is on the market in Europe. The Recover LP25 consists of a portable pump and a catheter that unloads the left ventricle. The device serves as an alternative to an intra-aortic balloon pump, but is not ECG-dependent.
FlowMedica (Fremont, California) exhibited the Benephit Delta Infusion System, a catheter-based system that provides direct administration of vasodilators and renal protective agents to the kidneys. The device can be used to treat fluid overload in heart failure patients, and to help prevent contrast nephropathy in the cath lab. About 15% of heart failure patients have cardio-renal failure, and can benefit from treatment with the FlowMedica device. Overall, the company estimates that 650,000 patients in the U.S. alone could benefit from treatment with the Benephit system for the two initial target indications. The product was cleared for marketing by the FDA in mid-March.
There also is continued growth in utilization of left-ventricular assist devices, including the HeartMate II from Thoratec (Pleasanton, California), the DeBakey VAD from MicroMed Technology (Houston) and the Novacor LVAD from World Heart (Oakland, California). At an ACC session on LVADs, the latest results of studies of the relative benefits of continuous flow assist devices such as the DeBakey VAD vs. pulsatile devices such as the HeartMate and Novacor VADs were described. There is a clear reduction in left ventricular diastolic and systolic volume and mass, and an increase in left ventricular ejection fraction (EF), when either type of device (continuous or pulsatile) is implanted, but the reduction in volume and increases in EF are greater with a pulsatile device such as the Novacor LVAD vs. a continuous flow device, indicating that pulsatile devices provide a greater degree of unloading. Improved tissue morphology is observed with both types of device, but for the sickest patients, a pulsatile flow device is preferred because greater improvement in the pumping efficiency of the heart is achieved.
Niche focus for ancillary devices
Other segments of the percutaneous device market with applications in cardiology include embolic protection devices and thrombectomy devices. Both segments have experienced some setbacks recently because of negative results of trials that evaluated their efficacy in improving outcome for myocardial infarction patients. Results of the PROMISE trial, which assessed the use of the FilterWire from Boston Scientific (Natick, Massachusetts) in myocardial infarction (MI) patients, were presented at the ACC sessions. Contrary to the expectations of many cardiologists, MI patients treated with the FilterWire exhibited no improvement in blood flow and no reduction in infarct size. The results are consistent with outcomes observed in the EMERALD trial using the PercuSurge GuardWire balloon protection device marketed by Medtronic (Minneapolis), as well as with results using throm-bectomy catheters such as the AngioJet from Possis Medical (also Minneapolis), and indicate either that none of the devices can prevent embolization in MI when used during reperfusion, or that distal embolization is not as important in determining outcomes in patients who are reperfused following an MI as is commonly believed. In addition, for the AngioJet trial there was a trend toward an increase in death rate for MI patients who were treated with the device vs. controls, although the death rate for controls was considerably lower than expected. Nevertheless, the results have led to a recommendation that the AngioJet only be used in MI patients with visible thrombus.
The negative results with embolic protection and coronary thrombectomy devices in myocardial infarct patients have dampened investor enthusiasm for the technologies. For example, Pathway Medical (Redmond, Washington), developer of the CardioPath catheter for removal of thrombus and atheroma, now is focusing most of its effort on debulking in peripheral vascular disease, having shelved development of a coronary catheter for which studies had been completed in Europe once the negative results with the Possis device were reported. The Pathway catheter, a rotational atherectomy device, is, however, showing promise for removal of thrombus and calcific disease in the peripheral vessels, and will be evaluated in peripheral vascular trials in Europe starting later this year. Pathway said it expects to obtain 510(k) clearance in the U.S. market by the end of this year.
Other applications in which protection and throm-bectomy devices continue to show promise include treatment of saphenous vein graft disease and treatment of carotid disease. Kensey Nash (Exton, Pennsylvania), for example, is continuing to evaluate its TriActiv balloon protection system for use in neuro (carotid) and saphenous vein graft disease, where distal protection devices have been shown to provide benefit.
Cell therapy/surgical treatment advances
The latest developments in cell therapy for the treatment of heart failure also were discussed at the ACC sessions. There is potentially a major new market opportunity for products used to deliver treatments such as stem cell therapy to patients with heart failure. So far, however, studies have produced mixed results. While some studies have shown moderate benefits, most have been designed to assess safety and not to produce maximum therapeutic effect. In addition, only a few companies have invested to develop optimized devices for harvest, culture and targeted delivery of cells to the heart. As discussed by A. Terzic, MD, of the Mayo Clinic (Rochester, Minnesota), at a satellite symposium sponsored by CV Therapeutics (Palo Alto, California), three types of stem cell therapy are under development for cardiac applications, including embryonic stem cell therapy, stimulation of growth of heart progenitor cells resident in the patient, and treatments using autologous adult progenitor cells harvested from other sites in the patient such as the thigh muscle.
Studies conducted at the Mayo Clinic have shown that cells and tissues having cardiac characteristics, such as cardiac-specific molecular tissue markers, electrical activity, and tissue striation, can be produced in vivo, but the process requires tight control. If an excessive dose of cells is injected in an attempt to achieve maximum therapeutic effect, large tumors can form based on results of animal studies. The effect is related to the therapeutic index of the injected material, i.e., the percentage of injected cells capable of differentiating into cardiac cells once injected into the heart. Terzic has conducted an extensive research program to develop biostimulation and cell selection methods to improve the therapeutic index, including proteomic studies to identify cardiogenic factors, identifying the steps involved in generating cardiac tissue, and development of culture techniques that produce large quantities of differentiated cells. Terzic also is employing treatment of the cultured cells with tumor necrosis factor alpha (TNF-?) to prevent tumorigenesis. In the most recent animal studies, Terzic has achieved larger areas of engraftment, and observed myocardial repair as well as improvement in mechanical function of the anterior wall of the heart.
Devices for improving the accuracy of delivery of cell preparations to infracted tissues will also be important for development of successful cell transplant therapy for heart failure. Numerous studies have used the NOGA guidance system and the BioSense injection catheter from Cordis Endovascular (Miami Lakes, Florida) to guide the cell injection process. One recent advance is the introduction of a new technique for in vivo tracking of injected cells, developed by BioPhysics Assay Laboratory (Worchester, Massachusetts). The method involves nanometer-sized colloids that can be quantified by neutron activation and imaged by fluorescence and MRI.
Cell labeling involves a simple incubation of the cells with the colloid label, followed by a wash step to clear excess label from the cell preparation. In one configuration, the colloid is a non-radioactive material that remains in the cells after they are injected into a host, and which can be quantified via biopsy and neutron activation analysis. Other colloids incorporating gadolinium allow non-invasive in vivo cell tracking via MR imaging, making it possible to follow the course of therapy in real time. Development of devices for use in cell therapy is challenging because of the need to deal with sterility issues throughout the cell isolation, culture, and delivery process; difficulty in harvesting and culturing an adequate number of cells with a sufficiently high therapeutic index; issues regarding potential side effects of stimulating agents used to enhance and guide cell differentiation; and the possibility of arrhythmias due to imperfect electrical integration of the implanted cells with the host heart tissue.
A number of researchers are focusing on the use of stem cells derived from the patient's bone marrow because technologies for sterile harvesting and delivery of such preparations already are available. In the BOOST trial involving 60 patients treated with bone marrow-derived stem cells, delivered using a PTCA catheter, an increase in ejection fraction was observed versus controls. Limitations of the bone marrow procedure include the need for multiple painful punctures to aspirate an adequate amount of marrow cells, the need for purification of stem cells prior to injection, and the small number of stem cells in bone marrow. However, the technique has exhibited the best efficacy of all the methods evaluated so far.
Another study, described by Nabil Dib, MD, of the Arizona Heart Institute (Phoenix), involved the use of autologous myoblast transplantation using cells derived from the patients' quadricep muscles. The study is the first multi-center U.S. trial to evaluate autologous myoblast transplantation. There were no adverse effects of the treatment, and an improvement in ejection fraction was observed from 22.9% to 34.6% over a 24-month follow-up period.
Researchers still are searching for the optimum cell type to use for restoration of damaged myocardial tissue, and for methods to improve the therapeutic index of the delivered cells in order to produce a significant and durable effect. It is likely that a considerable amount of research will be needed in order to understand the cellular processes involved in tissue restoration, and to develop methods to control those processes to obtain the desired effect.
The surgical treatment of heart disease also is changing as a result of the impact of minimally invasive technologies, both those that provide alternatives to surgical intervention (e.g., coronary stents) as well as minimally invasive surgical methods for performing bypass. As shown in Table 3 below, percutaneous techniques have become the dominant form of treatment used for coronary revascularization in the U.S., and similar trends have been observed in other countries such as Germany. As indicated by the data in Table 3, the conversion to percutaneous technology has shown some evidence of reaching a peak based on the most recent available data, although that data does not cover most of the period following the introduction of drug-eluting stents.
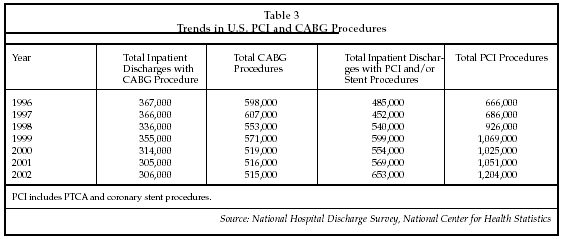
Statistics from the Mayo Clinic, however, indicate that there is in fact some recovery of procedure volume for cardiac surgery, with procedures increasing somewhat in the past year. Some 43% of procedures at the Mayo Clinic were performed for valve repair, 29% for CABG, 15% for repair of congenital heart defects, and 13% for other indications. In the future, coronary bypass surgery, as well as valve surgery, must become less invasive in order to remain a viable alternative to percutaneous technologies, except for patients with highly complex multi-vessel disease.
Experts presenting at the conference predicted that technologies such as TECAB (totally endoscopic coronary artery bypass), allowing avoiding of cardiopulmonary bypass; intraoperative guidance systems; and devices for performing automated anastomosis will be important tools in cardiac surgery, along with advanced imaging technologies, particularly MRI. Nevertheless, coronary bypass procedures in the U.S. are projected to drop from 290,000 in 2003 to 270,000 by 2008, continuing a trend toward greater use of PCI for revascularization. The impending introduction of percutaneous techniques for correcting heart valve defects as well as some congenital heart defects is likely to further erode the position of the cardiac surgeon in the treatment of heart disease.
