BBI Contributing Editor
SALT LAKE CITY, Utah This marked the first year that the leading professional organization for interventional radiology, the Society for Interventional Radiology (SIR; Fairfax, Virginia), convened under its new name. Formerly known as the Society for Cardiovascular and Interventional Radiology, the society's members perform a wide range of interventional procedures to treat peripheral artery and visceral disease. The products used by interventional radiologists comprise a market totaling $505 million in the U.S. in 2002, growing at about 17% per year, according to suppliers in the market.
The size of the market has attracted participation by a number of the largest suppliers in the medical device industry, while the market's diversity continues to attract a variety of small entrepreneurial ventures. Among the areas of growth for the interventional radiology profession as well as for suppliers of interventional radiology products are procedures and associated devices for the treatment of venous insufficiency, vascular aneurysms, cancer, and uterine fibroids.
A newly emerging opportunity highlighted at the SIR conference is cell transplant therapy, which builds on the developing field of tissue engineering and promises to provide important benefits for patients with diseases such as diabetes and possibly a wide range of other tissue degeneration disorders.
New developments also were described at the SIR conference in the areas of chronic total occlusion treatment, which has represented one of the major conditions resulting in referral of patients for surgical treatment. Devices to help prevent stroke are another focus for interventional radiology, including vena cava filters and carotid stents. The continued growth in prevalence of end-stage renal disease, and the accompanying growth in demand for renal dialysis, represents another expanding area of the market, comprised of devices for dialysis access and their maintenance. The market is expected to experience continued volume-driven growth for the foreseeable future due to aging of the population and continued increases in prevalence of diabetes, venous insufficiency, peripheral vascular disease and cancer.
Advances in treating venous insufficiency
Venous insufficiency, and its most common associated symptom of varicose veins, is one of the most prevalent diseases in the U.S. population. As shown in Table 8, the condition affects more than 50 million individuals in the U.S. to some degree. More than 10 million of those afflicted have visible varicose veins, and an estimated 4 million have chronic venous insufficiency. In more advanced stages, the condition can cause significant pain, swelling and skin ulcers that, if not treated, can progress to serious infection. Varicose veins result from reflux of blood in the veins when the one-way valves in the vein fail, causing blood to pool in the veins rather than being returned to the heart. Until recently, surgical stripping of varicose veins was the only available therapy, and typically was reserved for the most severe cases since most vascular surgeons do not consider vein disease to be worthy of surgical therapy, preferring to limit their practice to arterial repair. Vein disease is three- to four-fold more common than arterial disease, however, so there is a large number of patients who could benefit from treatment.
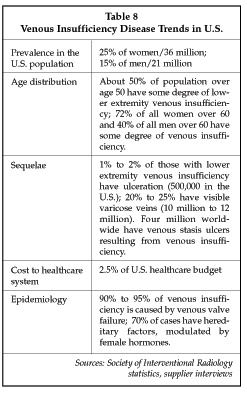
Even with the relatively low level of utilization of surgery for venous insufficiency, suppliers in the market estimate that more than 1 million persons worldwide undergo vein stripping procedures annually. Now, a number of new endoscopic and endovascular tools have become available for treating venous insufficiency, making therapy more attractive to patients, and creating opportunities for other specialists including interventional radiologists. Table 9 below describes devices available in the U.S. marketplace, as well as development-stage devices used for the treatment of venous insufficiency.
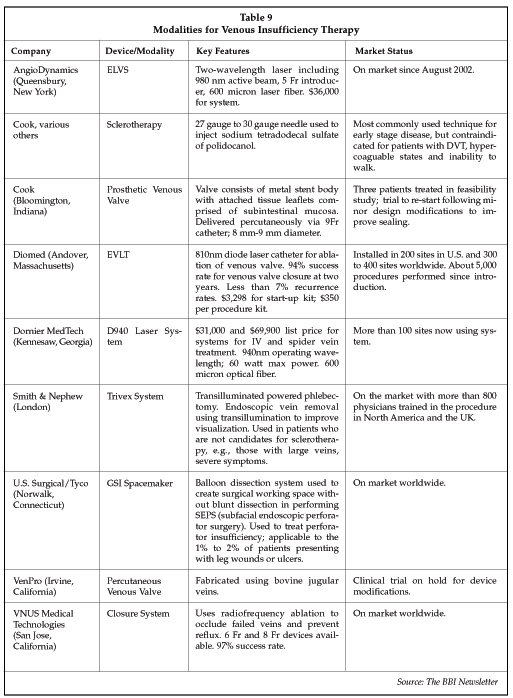
Four technologies are now available in the market, a segment that is estimated at about $7 million to $10 million (for less invasive devices) in 2002. The first less-invasive technology to be introduced for venous insufficiency treatment employs radiofrequency energy. Developed by VNUS Medical Technologies (San Jose, California), the Closure RF system uses a catheter to deliver RF energy to the vein. RF does not directly ablate the vein, but instead produces a reduction in lumen diameter by about 65%, as a result of heating of the catheter tip to 85 to 90 C. The narrowed diameter results in clotting of the vein and elimination of blood flow. More than 10,000 procedures have been performed worldwide with the Closure system since its launch in 1998. The product was cleared for marketing in the U.S. in March 1999.
A second minimally invasive alternative is laser treatment for varicose veins. Three companies now supply laser-based systems that allow percutaneous ablation of varicose veins, employing high-intensity light of 810 to 940 nanometers in wavelength. Prices for the systems range from about $25,000 and up, although one supplier places the system for a small start-up charge and allows physicians to pay on a per-procedure basis. Laser systems have proven effective for the treatment of venous insufficiency, with typical success rates of over 90% at two years. The cost of the procedure varies, depending on the region of the country and the payer, and ranges from about $1,900 to $3,000 per leg treated. Thousands of procedures have been performed with laser systems worldwide. The Diomed (Andover, Massachusetts) EVLT system has captured the largest share of the laser treatment market, with about 5,000 procedures performed worldwide since the product was introduced in January 2002. Competing laser systems launched more recently are now also gaining share in the market.
Perhaps the most innovative solution to venous insufficiency, however, is to replace the failed venous valves with prosthetic devices that restore normal blood flow patterns to the venous system. Cook (Bloomington, Indiana) is developing a prosthetic venous valve in collaboration with researchers at the Dotter Institute (Portland, Oregon) that shows considerable promise. The device employs subintestinal mucosa (SIS) for valve leaflets, a biologically derived material, and is delivered via percutaneous techniques. Valve function is immediately restored to the affected vein. Over time, cells from the patient populate the leaflet material, and the SIS biodegrades, leaving only the stent framework as a foreign material in the body. Following successful animal studies, three patients have been treated in British Columbia with all three exhibiting good results, including one with an ulcer that healed following the procedure. Trials are to resume shortly following some minor modifications to the device. VenPro (Irvine, California) also is developing a prosthetic venous valve for use in the treatment of venous insufficiency, in partnership with Percutaneous Valve Technologies (Fort Lee, New Jersey). The VenPro bioprosthesis is made from bovine jugular veins. Two versions are under development, a percutaneously implanted prosthesis and a surgically implanted version. The market opportunity is substantial, given the high prevalence of venous insufficiency.
The treatment of venous insufficiency represents a significant opportunity for interventional radiologists to expand their practice. As discussed by Gerald Niedzwiecki, MD, of Mease Countryside Hospital (Safety Harbor, Florida), president of Advanced Interventions, at the SIR meeting, physicians can net between $985 and $2,033 per procedure, depending on whether the procedure is performed in the hospital or in a private office, and also depending on the type of reimbursement available (Medicare being the lowest and cash-paying patients being the highest). Capital equipment costs can be recouped by performing as few as 26 procedures. Continued market expansion will be driven to a significant extent by the degree to which interventional radiologists are proactive in informing referring doctors and patients about the availability and benefits of the procedure.
Embolization therapies competition grows
Another interventional radiology procedure that represents a significant opportunity for expansion is transcatheter embolization. Devices for embolization therapy for aneurysms, particularly for cerebral aneurysms, represent a well-established market for suppliers, totaling about $20 million in the U.S. in 2002 and significantly more worldwide. A range of technologies are employed for embolization therapy, including metal coils delivered via catheters as well as particles of various types. The Target unit of Boston Scientific (Natick, Massachusetts) remains the leader in the coil segment, but new suppliers, including Micrus (Sunnyvale, California), have emerged and have succeeded in penetrating the market. The Micrus coils provide rapid (3 to 3.5 second) deployment, and the company has just launched the UltiPaq, a small, flexible coil used to complete filling of an aneurysm after initial occlusion with the larger space-filling MicruSphere coil. The UltiPaq coils now represent about 50% of all coils implanted for Micrus.
A new application, uterine fibroid embolization (UFE), has recently emerged that has created an opportunity for new entrants in the market. UFE is typically performed with particles rather than coils, and provides an alternative to surgical treatments such as hysterectomy or myomectomy that is minimally invasive and less expensive. The results of a study presented at the SIR conference by M. Garcia, et al., of Christiaria Care Health System (Newark, Delaware), showed that 92% of patients treated with UFE had complete resolution or significant improvement in symptoms at one-year follow-up, with 96% of those requiring no further therapy.
The current leader in the UFE device market is BioSphere Medical (Rockland, Massachusetts), which claims an 80% share of the market for transcatheter embolization particles (i.e., excluding the embolization coil segment). BioSphere entered the U.S. market only three years ago and has quickly established its EmboGold microspheres as the preferred technology for UFE. EmboGold microspheres have a uniform cross-sectional diameter, simplifying the task of selecting the proper particle size to achieve optimal embolization and avoiding clumping of particles that can cause catheter occlusion with other types of embolic materials. Insurers including Blue Cross now reimburse for UFE, and the BioSphere particles are FDA-cleared for use in the procedure. The key remaining hurdle to more widespread use of UFE for uterine fibroids is lack of awareness of the availability of the treatment on the part of patients as well as the ob/gyns who typically refer women for the procedure. BioSphere has mounted an aggressive marketing campaign, including funding of a company that provides training in the procedure, to help drive more widespread adoption.
A study presented at the SIR conference by James Spies, MD, of Georgetown University Hospital (Washington), found that UFE is as effective as hysterectomy in resolving common symptoms of uterine fibroids, but is associated with a shorter hospital stay (0.8 days vs. 2.3 days), quicker return to work (10.7 days vs. 32.5 days), a more than 50% reduction in blood loss and fewer complications (27.5% vs. 50%) as compared to women undergoing hysterectomy. Awareness of the benefits of UFE now appears to be growing based on the results of another study of gynecologists' opinions of the technique, as reported by E.K. Arleo of Yale University (New Haven, Connecticut) at the SIR conference. A poll of 21 women who were asked to describe the opinions of their gynecologists regarding UFE now vs. two years ago found that the number opposing the procedure dropped from 76% to 33%. Furthermore, the percentage of gynecologists who offered UFE as a treatment alternative rose from 5% to 38% over the two-year period. Although the sample is quite small, the data is consistent with adoption trends observed by suppliers. The Society of Interventional Radiology also has been a strong proponent of greater use of the UFE procedure as an alternative to hysterectomy. Other suppliers of embolization particles used in UFE procedures include Cook (PVA particles) and Boston Scientific (Contour SE particles).
A unique new approach to performing UFE was described by researchers from InSightec-TxSonics (Dallas, Texas). Insightec is a venture formed by GE Medical Systems (Milwaukee, Wisconsin) and Elbit Medical Imaging (Haifa, Israel) in 1999. The company is developing the ExAblate 2000 High Intensity Focused Ultrasound (HIFU) system for applications in breast cancer treatment and uterine fibroid ablation. A study described at the SIR conference involving 16 women with uterine fibroids found that 88% had improved symptoms after HIFU therapy, and none experienced significant pain or discomfort during the procedure. That finding contrasts with the experiences of patients treated with particle embolization, who often experience severe pain during the procedure. All of the patients in the Insightec study were treated on an outpatient basis.
Chemoembolization is another important and growing application for interventional radiology techniques in the treatment of cancer. Key applications include treatment of liver tumors, head and neck tumors, and colorectal tumors. The same types of particles used for UFE are used in chemoembolization, but in addition chemotherapy drugs are infused at the tumor site. The technique both cuts off the flow of blood to the tumor and allows high doses of chemotherapy agents to be delivered locally at the tumor site while avoiding systemic exposure to the drug. As a result, adverse effects of chemotherapy are minimized and killing of tumor cells is maximized. Treatment can also be performed without using chemotherapy drugs, typically to ablate as much of the tumor as possible prior to surgical resection, reducing post-surgical morbidity. Studies presented at the SIR conference show that combining embolization therapy with systemic chemotherapy has added benefits, as that allows occult micrometastases to be ablated along with the primary tumor.
Radiation therapy can also be delivered locally or regionally via particles similar to those used in chemoembolization. Microparticles such as the Theraspheres from MDS Nordian (Kanata, Ontario) and SIR-Spheres from Sirtex Medical Ltd. (North Ryde, Australia) are examples of radioactive microspheres used for radiation treatment of cancer. Theraspheres are cleared by the FDA for humanitarian use in the treatment of inoperable hepatocellular carcinoma, and SIR-Spheres are cleared for the treatment of colorectal hepatic metastases. Both contain yttrium-90 and are introduced via catheterization of an artery supplying blood to the target tumor. The spheres are 20 microns to 30 microns in diameter, and the radioactive material has a short half-life of two to three days, with an effective therapeutic range of 2.5 mm. A vial of microparticles suitable for use in a single treatment (e.g., Therasphere) costs about $10,000.
Radioactive embolization particles have proven to be a very effective cancer treatment modality in trials. For example, a randomized trial using SIR-Spheres plus systemic chemotherapy showed a 73% response rate vs. 0% in controls treated only with systemic chemotherapy, with a three-fold increase in time-to-disease progression. Toxicity remained acceptable, although about two-fold higher than in patients treated with chemotherapy. A study conducted with Theraspheres in colorectal cancer also showed promising results, with a 90% response rate in patients treated with Theraspheres vs. a 20% rate in patients treated with conventional chemotherapy. The clear advantages of chemoembolization therapy have created growing interest in the technology, and Sirtex is now the subject of an acquisition offer by Cephalon (West Chester, Pennsylvania), a developer of cancer treatment drugs.
Transcatheter techniques also can be used to deliver cells, using an approach similar to that used for delivery of embolization particles. A major advance in diabetes therapy using interventional techniques for islet cell implantation was described by Richard Owen, MD, of Alberta Hospital (Edmonton, Alberta), at the SIR conference. Islet cell implantation to treat diabetes has been under development since the late 1970s, initially using surgical implantation. More recently, techniques for transplantation using microinfusion catheters have been developed. The team led by Owens has treated a total of 48 diabetic patients over the past three years, performing 90 cell implant procedures using a microcatheter delivery system from Cook Canada. Islet cells are isolated from the pancreas of a blood-matched organ donor. In most cases, two organs are required in order to obtain a sufficient number of islet cells, and more than one transplant procedure is typically required per patient. On average, only about 5,000 islet cell equivalents per kilogram can be obtained from one donor pancreas. In the study, all of the patients who received more than 9,000 islet cell equivalents per kilogram (857,000 total cells) became insulin-independent. Prior to treatment, all patients had uncontrolled blood glucose levels. At one year, 81% of the treated patients remained insulin-independent, and three of four patients treated over three years ago continue to be insulin-independent. Even those patients who develop a new requirement for insulin have improved blood glucose control.
The procedure involves insertion of a needle into the portal vein under X-ray or ultrasound guidance. No anesthesia is required. The major complication rate for the study conducted by Owen was 9%, with complications including intra-abdominal hemorrhage and portal vein occlusion. One approach to minimizing hemorrhagic complications is to embolize the access tract with agents such as Gel-Foam, a product manufactured by Pharmacia (Peapack, New Jersey), which in April was acquired by Pfizer (New York). Patients must take immunosuppressive drugs after the procedure to avoid rejection of the implanted cells. With prior-generation immunosuppressive agents, there was a significant amount of damage to the implanted islet cells, greatly limiting the effectiveness of the procedure. But the latest-generation drugs do not cause as much damage to the implanted cells as previous versions, one of the factors that has increased the success rate of the procedure. Nevertheless, immune rejection of the transplanted cells remains the primary cause for failure of the procedure. At present, the islet cell implantation procedure is reserved for only a small percentage of the estimated 100 million diabetics worldwide, namely insulin-dependent (Type 1) diabetics with uncontrolled glucose levels, and use is limited by the supply of donor organs from cadavers. However, ongoing efforts to produce islet cells from autologous (patient-derived) stem cells could eliminate that latter restriction in the future, as well as helping to avoid immune rejection and the necessity for taking immunosuppressive drugs. The size of the market opportunity remains a question for suppliers, since relatively inexpensive devices are used in the cell implant procedure.
Growing market in dialysis access
Another growth segment within the interventional radiology device market is products for dialysis access. As shown in Table 10, the number of patients undergoing hemodialysis is continuing to increase. That trend holds true both in the U.S. and worldwide. There are 6 million people in the U.S. with some degree of renal disease, and 300,000 with high creatinine levels who are progressing to chronic kidney disease and the need for dialysis therapy. About 72% of hemodialysis patients start treatment using a hemodialysis catheter, and subsequently switch to either an arteriovenous fistula or a synthetic graft for dialysis access.
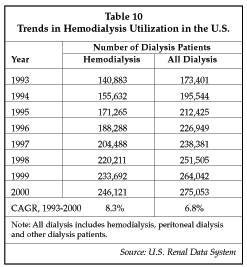
Although there has been a major effort by the U.S. government to stimulate the use of native fistulas for dialysis access, with a goal of using fistulas in 50% of all dialysis patients, the generally poor condition of the blood vessels in the diabetic population often makes use of a fistula impossible. Only about 27% of dialysis patients in the U.S. have native fistulas. An additional 20% use hemodialysis catheters, and the remaining 53% have prosthetic grafts. Unfortunately, prosthetic grafts fail due to stenosis at the graft anastomosis and thrombosis, with a seven-fold higher failure rate for prosthetic grafts vs. native fistulas. Typical patency rates for ePTFE grafts used for dialysis access are 62% to 83% in the first year, 50% to 77% at two years, and rates are progressively lower for longer periods. Arteriovenous fistulas typically have longer patency, lasting up to eight years in some cases.
Costs to maintain patency are $75,611 for the first year and $54,555 for the second year for patients with grafts, vs. $68,000 in year one for patients with fistulas. On average, one to two interventions are required each year to maintain patency of a prosthetic dialysis graft. In spite of the advantages of native fistulas, however, it has proven difficult to increase their utilization because of the difficulty in creating a properly maturing fistula in many patients (e.g., diabetics, older patients, women), and because the condition of some patients does not allow them to delay starting hemodialysis.
Devices used to open thrombosed grafts and fistulas include high-pressure PTA balloon catheters, the Cutting Balloon from Boston Scientific and a variety of thrombectomy devices, including the Percutaneous Thrombectomy Device supplied by Arrow International (Reading, Pennsylvania), the Hydrolyser from Cordis Endovascular (Miami Lakes, Florida), the AngioJet from Possis Medical (Minneapolis, Minnesota), the XTD Thrombectomy System from Xtrak Medical (Salem, New Hampshire) and the Helix from ev3/Microvena (White Bear Lake, Minnesota).
Idev Technologies (Houston, Texas) markets the Akonya Eliminator mechanical thrombectomy device in Europe.
Another approach to clot removal is to use a surgically inserted catheter such as the Fogarty catheter or the Adherent Clot Catheter from Edwards LifeSciences (Irvine, California).
However, a number of new devices were exhibited at the SIR conference that may improve graft and fistula maintenance and capture a share of the growing market for such products. REX Medical (Radnor, Pennsylvania), for example, is developing a new device for clearing grafts that uses sinusoidal wire oscillating at 4,000 rpm to macerate and dissolve blood clots. The next generation of the REX device will add an angioplasty balloon and possibly a compliant balloon for plug removal on the oscillating wire catheter, allowing most procedural steps to be accomplished using a single device.
Two new thrombectomy devices, the Solera Plus Thrombectomy System and the Trellis Reserve device, were exhibited by Bacchus Vascular (Santa Clara, California). Bacchus Vascular was founded in 1999 by Dr. Thomas Fogarty to develop new devices for treating cardiovascular and peripheral vascular occlusive disease. The Solera Plus is designed for use in clearing clotted dialysis grafts and uses a rotating macerator contained in a stationary protective basket (0.018" guidewire compatible) at the tip of a catheter. A hand-held drive unit is included with a Luer fitting to allow attachment of a syringe for aspiration of the macerated thrombus. The entire device, including the drive unit, is disposable. The Trellis device provides the capability to use thrombolytic agents for clot dissolution with higher efficiency and reduced risk of hemorrhage. The Trellis Reserve uses distal and proximal occlusion balloons and an oscillating dispersion wire to help disperse an infused thrombolytic agent throughout the clot. Recommended treatment time is 15 minutes, and the treatment area can be aspirated, if desired, after clot lysis. The system has a 6 Fr uninflated profile, allowing percutaneous insertion. Use of the occlusion balloon allows higher doses of thrombolytic agents to be infused without the risk of inducing a hemorrhage. A poster presentation from Bacchus demonstrated that there is no leakage of the thrombolytic agent around the deployed balloons.
The completely disposable device is priced at $1,495, with no capital equipment required. The company has calculated that the device may save money for the hospital, however, when compared to conventional thrombolysis, because of its ease of use and rapid treatment time, as well as elimination of ICU costs. Typical savings including the cost of the Trellis device are estimated at $1,500 to $2,500 per procedure. The primary application for the Trellis is treatment of critical limb ischemia, i.e., clearance of clots in the peripheral arteries.
New devices using ultrasound thrombolysis were exhibited at the SIR conference. OmniSonics Medical Technologies (Wilmington, Massachusetts) showed its Resolution System, which operates at 20 MHz and can be used in conjunction with other treatments such as thrombolytic agents to dissolve clots. Initial applications are for treatment of thrombosed hemodialysis access sites and clotted arteries in the lower extremities. The Resolution system is not yet cleared for marketing in the U.S., but has a CE mark allowing it to be sold in Europe. Ultrasound energy is emitted in a uniform 360 pattern around the Resolution wire, causing cavitation in the surrounding fluid that macerates clot into microparticles smaller than red blood cells. The wire has a maximum diameter of 0.030" and a 20 cm active length. Treatment times are quite short, with only about six minutes required to clear a chronic total occlusion and less for clearing of a thrombosed access graft. In addition to the disposable catheter, the system includes an ultrasound generator. Other development-stage ultrasound thrombectomy systems included the Acolysis system from Vascular Solutions (Minneapolis, Minnesota) and a system from AngioSonics (Morrisville, North Carolina).
Peripheral, visceral intervention technologies
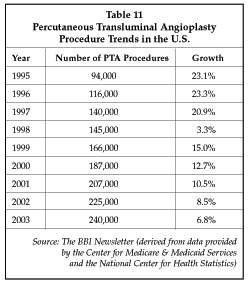
Interventional technologies for peripheral revascularization and for visceral interventions such as transjugular intraperitoneal portosystemic shunt (TIPS) procedures are also continuing to improve, helping to drive the continued movement towards increased use of minimally invasive methods versus surgical treatment. As shown in Table 11, the estimated number of percutaneous transluminal angioplasty (PTA) procedures is growing at about 7% per year, and the compound annual growth rate in procedure volume is expected to be about 8% over the 1999-2005 interval. Based on data from the Center for Medicare & Medicaid Services, the number of PTA procedures performed in the Medicare population increased at a 13.3% compound annual rate between 1995 and 2001, reaching about 187,000 in 2001. Renal/visceral and venous PTA procedures have exhibited the strongest growth.
One important trend that is helping to drive growth in peripheral vascular interventions is the development of new devices that show promise of providing improved results in applications that have proven challenging so far, such as treatment of femoropopliteal and renal stenosis. Cryoplasty is an example of a new revascularization technology with potential applications in femoropopliteal arteries, being developed by CryoVascular Systems (Los Gatos, California). Cryoplasty has shown considerable promise in clinical trials, with one study presented by Kettenbach, et al., of the University of Vienna (Vienna, Austria) showing a 100% patency rate at six months in a pilot study of seven patients. The device was used as the primary therapeutic modality, achieving an 85% procedural success rate, and no stents were required in any of the patients. The CryoVascular PolarCath also has been used successfully in the treatment of infrainguinal lesions and superficial femoral artery lesions. The CryoVascular system employs nitrous oxide to cool the surface of the dilatation balloon to -10 C. Researchers have shown that the freezing and thawing process causes apoptosis of smooth muscle cells, attacking the primary cell type involved in restenosis.
Boston Scientific's Cutting Balloon also is being used in peripheral vascular therapy, producing promising results in clinical studies of limb salvage. The growing penetration of interventional techniques for the treatment of occlusions in the peripheral vessels and particularly in critical limb ischemia is indicated by data discussed by Amman Bolia, MD, of Leicester, UK, at the SIR conference, who noted that 64% of patients with critical limb ischemia were treated with surgery prior to 1994. By 1997, as a result of the rapid adoption of improved interventional technologies, 64% of patients were being treated with interventional therapy, with equivalent outcomes.
Continued advances in interventional techniques, particularly for the treatment of infrapopliteal lesions, have continued to drive further replacement of surgical therapy over the past few years. For example, a team led by McNamara at the UCLA Medical Center (Los Angeles, California) used the Cutting Balloon to treat patients at risk for limb loss due to stenoses or occlusions and achieved an 85% limb salvage rate at one year, with a 33% restenosis rate at six months. While the restenosis rate appears high, it compares favorably to restenosis rates of up to 70% using standard PTA balloon catheters. Use of the Cutting Balloon also may avoid the need for stents, a desirable feature since existing stents do not perform well in the lower legs, particularly in below-the-knee applications. The Bacchus Vascular Trellis device has also been used in limb salvage, for the treatment of superficial femoral artery thrombosis leading to threatened limb loss. In that application the Trellis speeds up clearing of the clot, reducing the cost of treatment.
Another device that may help expand the range of lesions that can be treated successfully with interventional radiology techniques is the CrossPoint Catheter from TransVascular (Menlo Park, California). The CrossPoint is proving particularly useful in facilitating the treatment of chronic total occlusions in the legs. The device has dual lumens and incorporates IVUS technology from Jomed (Helsingborg, Sweden) for integrated imaging guidance during a procedure. The CrossPoint includes a deployable needle that can be used to help direct the guidewire into the subintimal space, allowing an occlusion to be bypassed, with the guidewire then redirected into the true lumen of the vessel beyond the blockage. Another device, the Extreme laser catheter from Spectranetics (Colorado Springs, Colorado), also is being used to treat total occlusions in the superficial femoral arteries and for treatment of critical limb ischemia. Two clinical trials, PELA and LACI II, are under way with the device. The excimer laser dissolves thrombus and ablates plaque, and, with proper sizing to match the catheter outside diameter to the vessel lumen, does not require a complex guidance mechanism.
Stents also are playing a role in the expansion of applications of interventional therapy in the peripheral vessels. Vascular Architects (San Jose, California) has introduced the aSpire Covered Stent, comprised of a nitinol frame configured in a spiral that is fully covered by ePTFE. The device is crush-resistant, an important characteristic for applications in the leg, and provides enhanced luminal coverage while simultaneously protecting side branches of the lumen. Covered stents have proven superior to conventional bare stents in applications such as SFA revascularization, where bare stents exhibit restenosis rates of 30% to 80% regardless of the stent used. The Hemobahn SG stent-graft from W.L. Gore (Flagstaff, Arizona) has been used with success in the SFA, achieving an 83% secondary patency at two years in long segments, results that are equivalent to those obtained with surgically implanted vascular grafts. The aSpire covered stent may provide added benefits because of its high radial strength. Another promising development-stage device in the category is the WallGraft from Boston Scientific, which is being evaluated for the treatment of aneurysms and trauma-induced lesions in the peripheral vessels. Stent-grafts represent one of the most rapidly growing segments in the interventional radiology market, with worldwide sales expected to exceed $380 million in 2003, with essentially all sales so far attributable to devices used for the treatment of abdominal aortic aneurysms.
A final area of development focus in interventional radiology is temporary and removable vena caval filters. Temporary filters, which are attached to a guidewire or catheter for removal, are usually left in place for only a few days, since the anchoring mechanism results in some restrictions on patient mobility and may serve as a source for infection. Retrievable filters avoid the limitations of temporary filters, and are used much like permanent filters but can be removed when there is no longer a requirement for protection against thromboembolism. Bard Peripheral Vascular (Tempe, Arizona) is one of the leaders in the development of retrievable filters. The Bard Recovery filter is approved in the U.S. and Europe for use as a permanent filter, but can be removed if desired for periods of months post-placement, based on results from trials conducted by physicians presenting at the SIR conference. The Recovery is based on the design of the Bard SNF filter, a nitinol device implanted with a 7 Fr delivery system. A 10 Fr urethane cone is used to recover the filter, which can be visualized via MR imaging.
As discussed by Murray Asch, MD, of Mount Sinai Hospital (Toronto, Ontario) at the SIR conference, the Recovery filter can be removed at up to 12 weeks after implantation, indicating that Bard has succeeded in significantly reducing incorporation of the filter into the surrounding vascular endothelium. Of 58 Recovery filters placed in the study conducted by Asch, 46 were removed, with an average placement time of 62 days. Eleven filters were removed that contained trapped clots, demonstrating that the device works as intended to protect against thromboembolism.
One advantage of removable filters is that patients with venous thromboembolic disease or malignancy can sometimes be misdiagnosed as requiring a filter, resulting in their living with a permanent implant that is prone to long-term complications. Retrievable filters resolve such problems, and have the added benefit that patients who only require short-term filter protection can be safely protected without risking long-term complications. Asch reports that patients also tend to be discharged earlier and return to work sooner, and generally have a more positive attitude about their recovery after the filter is removed.
