Chilling.
It isn’t often that a bit of news is a perfect fit to that one-word description, but the recent decision by a Los Angeles jury in the first case to be decided pitting Johnson & Johnson (J&J; New Brunswick, New Jersey) against what is a cast of thousands of litigants is nothing short of that.
Oh sure, there’s the usual post-decision posturing by the company per appealing the $8.3 million-plus jury award, but c’mon folks, the implications of this case for the medical-products giant – and truly for all other companies trying to do business in the med-tech space – are simply enormous.
Yes, the eventual financial onus will fall largely on the insurers who’ll be left holding the bag. It’s doubtful that many common folk will be weeping over that outcome – picture the folks in line at the phone company in the film Fun With Dick and Jane, bursting into cheers when D&J come in to rob the coffers – but the fact is that the outcome will make doing business as a device company even more difficult.
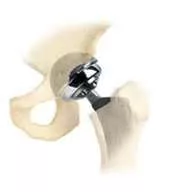
First of all, consider the scope of the entire J&J/DePuy Orthopedics hip implant legalities. This was the first of thousands of cases, with something in excess of 10,000 lawsuits pending over the Articular Surface Replacement, or A.S.R. The fiscal gymnastics of taking $8.3 million and multiplying it by that number of cases would fry even a Cray supercomputer. With thousands of the individual cases already having been consolidated into a single proceeding in a federal district court in Ohio, we wouldn’t expect to see every victorious litigant capture $8 million for pain and suffering as did the first California winner, but the number of digits following the dollar sign is still likely to be breathtaking.
Secondly, there’s the trust thing – from both the public (consumer) and user (surgeon) perspectives. The A.S.R. was sold in the U.S. and elsewhere in the world for years even though J&J’s own internal documents, as shown during the Los Angeles trial, clearly indicated DePuy knew there were problems with the metal-on-metal design.
Surgeons apparently told the company with great regularity that problems with metallic debris made it clear sales of the device should be slowed or stopped, but that didn’t happen until 2010, with sales of the A.S.R. continuing throughout much of the prior decade.
Even in a country where litigation of all sorts occurs pretty much at the drop of a hat, and where makers (and users) of medical products are among the most favored of targets, the scope of the J&J/DePuy cases is just plain startling.