The capital markets cooled off somewhat in November with the Nasdaq and Dow Jones Industrial averages both finishing with small gains of 2.5 percent and 3.4 percent, respectively. The uncertain market conditions were also enough to halt the initial public offering (IPO) aspirations of several biotech companies who pulled their IPOs this month. Those included Celladon Corp., of San Diego, which has a heart failure compound Mydicar in Phase II trials; Globeimmune Inc., of Louisville, Colo.; Tetralogic Pharmaceuticals Inc., of Malvern, Pa.; and Vital Therapies Inc., of San Diego.
Xencor Inc., of Monrovia, Calif., a biotech developing antibodies for severe autoimmune/allergic diseases and cancer, tried hard to get its IPO out of the door by filing an amended S-1, slashing the price of its proposed offering from a range of $14 to $16 to $7 per share while upsizing the shares from 5 million to 10.7 million in an effort to generate proceeds in the same ballpark of approximately $75 million. Unfortunately, the move didn’t bear fruit with the company halting the IPO, citing poor market conditions. (See BioWorld Today, Nov. 18, 2013.)
Window Remains Open
The biotech IPO “window” hasn’t closed, however. In fact, there were two successful biotech IPOs, which bookended the month.
Natick, Mass.-based Karyopharm Therapeutics Inc. became the 34th biopharma firm to price an IPO in the U.S. this year, according to BioWorld Snapshots. The company generated $108.8 million in an upsized offering by increasing the number of shares offered from 5.7 million to 6.8 million, pricing them at $16 each, at the high end of its proposed range. Karyopharm filed an S-1 under the Jumpstart Our Business Startups Act, originally seeking to raise $80 million. Proceeds from the IPO will go toward the firm’s pipeline of selective inhibitors of nuclear transport (SINE) compounds, which are designed to inhibit the nuclear export protein XPO1. Lead SINE compound selinexor (formerly KPT-300) is in Phase I testing in pre-treated relapsed and/or refractory hematological and solid tumor cancers, with plans to move into Phase II by early 2014. The company’s pipeline also includes an inflammatory disease candidate, KPT-350, which is in preclinical development. A SINE compound related to selinexor is in pivotal studies to treat lymphomas in dogs. (See BioWorld Today, Nov. 7, 2013.)
By the time Redwood City, Calif.-based Relypsa Inc. initiated its offering, the tougher market conditions were beginning to exert their effects. The company had to drop its share price twice from a range of $16 to $19 down to $12, and finally to $11, which got the deal done with 6.85 million shares sold. The company said it expected approximately $67.4 million in net proceeds from the offering before overallotments, with 28.7 million shares outstanding after the offering. (See BioWorld Today, Nov. 18, 2013.)
Interestingly enough, Relypsa has performed very well since its debut with its share value jumping 40 percent. On the other hand, Karyopharm’s share price, although it jumped to $19 on debut, it has fallen since then to close where it started at $16 by the market close on Nov. 26.
Despite the fact that the enthusiasm for biotech IPOs has waned somewhat, no matter what the final month of the year holds, it still will be a record-setting year for those new issues with approximately $2.8 billion raised so far by the 2013 IPO graduates, according to BioWorld Snapshots.
Those newly minted public biotech companies have taken advantage of welcoming capital markets with investors eager to jump on board. That positive environment also has swept biotech valuations to new heights with the 259 publicly listed companies tracked by the BioWorld Stock Report recording an average 40 percent increase in their share values year to date.
To emphasize what an incredible year it has been, BioWorld’s Blue Chip Index, a price-weighted index that includes 21 of the top biotechnology companies has recorded a 76 percent increase in value, in the same time period and has considerably outpaced the general markets with the Dow Jones Industrial average recording a 22 percent YTD increase and the Nasdaq Composite index a 33 percent increase. (See BioWorld Blue Chip Index, below.)
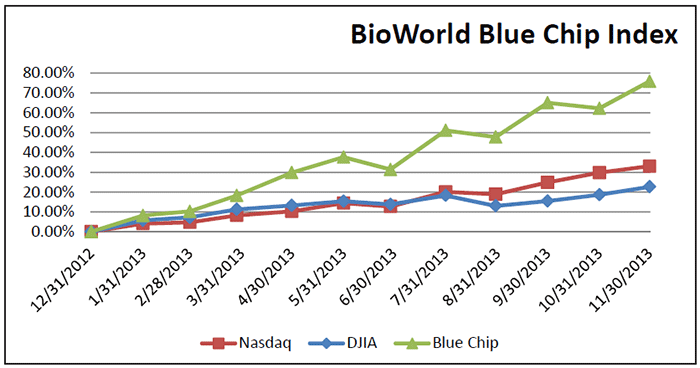
Same Story
It has been the same story for companies comprising the BioWorld Growth Index propelling its value by 106 percent so far this year. (See BioWorld Growth Index, below.)
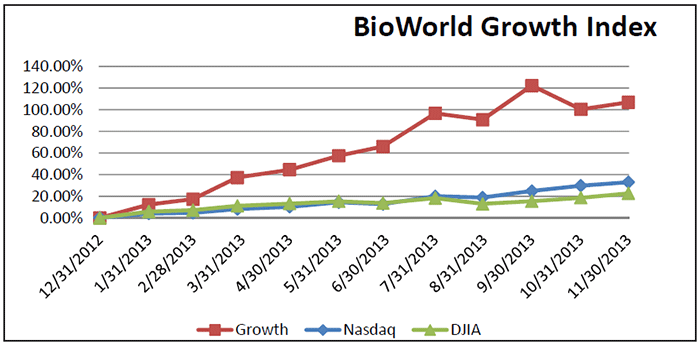
November was also a good month for the group of public biotech companies comprising BioWorld’s Emerging Biotech Index, which recorded a 10 percent increase in value (64 percent YTD). The index outpaced both the BioWorld Blue Chip and the BioWorld Growth indices, which recorded increases of 8 percent and 3 percent, respectively. (See BioWorld Emerging Biotech Index, below.)
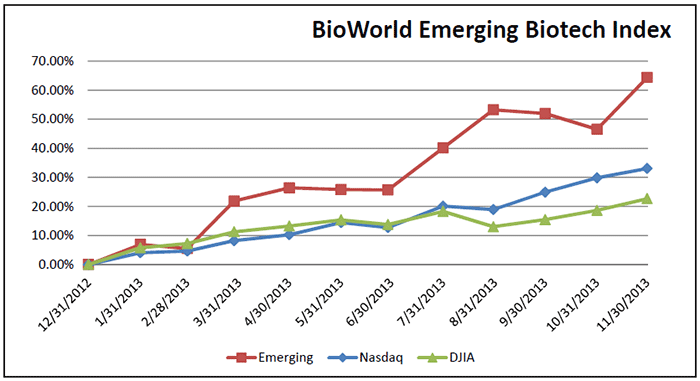
Leading the gainers in the group was Orexigen Therapeutics Inc. whose shares closed the month up 31 percent. The company created a lot of market buzz with early results from the Phase III trial known as the Light study for its obesity drug Contrave (naltrexone/bupropion). (See BioWorld Today, Nov. 26, 2013.)
In an interim analysis, the trial met pre-specified criteria to exclude a hazard ratio of 2, using the upper limit of the 95 percent confidence interval, for excess risk of major adverse cardiovascular events in patients given Contrave as compared to placebo.
Sangamo Biosciences Inc., of Richmond, Calif., also had a good month, with its shares jumping by 26 percent. The company presented results of a Phase I trial of its gene therapy, CERE-110, for Alzheimer’s disease, showing that surgical delivery of the drug to the brain resulted in long-term expression of bioactive nerve growth factor and apparent stabilization of brain cell metabolic activity, as determined by PET-scans measuring glucose use. The product was well tolerated at every dose level.
With just one month to go before the end of 2013, the positive trends of our indices still indicate that public biotech companies developing therapeutics remain well on track for a record-setting year.
