BB&T Contributing Editor
DUSSELDORF, Germany – Healthcare has historically been centered in the hospital in Europe, with services provided by a network of approximately 15,600 institutions in Western and Eastern Europe, according to data from the World Health Organization (WHO; Geneva, Switzerland). A study from WHO's European Observatory on Health Care Systems found that hospitals account for 50% of the healthcare budget in many western European countries and 70% or more in countries of the former Soviet Union.
Within the past few years, however, driven by demands for reduced cost of care and improved efficiency, healthcare has begun to expand outside the traditional hospital setting with increased utilization of technologies such as point-of-care testing, minimally invasive surgery, remote patient monitoring and telemedicine. More sophisticated care is being delivered in the physician's office in Europe.
Other advances in healthcare technology impacting the market in Europe include regenerative medicine, new technologies for detecting chronic diseases such as diabetes, metabolic syndrome and cancer, including genetic testing, and advances in treatments for orthopedic disease.
The largest forum for introduction of new medical technologies, the MEDICA exhibition held here in mid-November, provided a window on the latest developments in the medical device market in Europe, as well as in the global market.
The current economic slowdown has not yet impacted marketing and business development activity in the medical device sector in Europe, at least as indicated by attendance figures for the MEDICA exhibition. The number of exhibitors and attendees was equivalent to the previous year's event, with a total of 4,313 companies exhibiting and 137,000 attendees at MEDICA and the companion COMPAMED exhibition.
Given the rapidity of the onset of the downturn, it is possible that manufacturers have not had time to respond to the changing environment, although most of those interviewed at MEDICA indicated that the medical device sector may prove to be less affected by trends in the global economy than other segments.
However, capital equipment vendors are likely to see some negative impact going forward since financing plays a key role in that segment of the market, particularly in the developing countries. The decline in the financial markets is, on the other hand, stimulating some medical products companies to re-evaluate their business structure.
For example, Cardinal Health (Dublin, Ohio) is planning a restructuring in response to shareholder pressure to maximize value that will result in a spin-off of its infection prevention, respiratory care, medication and supply dispensing, medical diagnostics, surgical, and Alaris drug delivery businesses as a separate public company, leaving the medical products distribution unit as part of Cardinal along with its pharmaceutical distribution business. The transaction, announced publicly in late September, is scheduled to be complete this June. A name for the spin-off company, which will be headquartered in San Diego, will be announced next month.
Another transaction reporteded during the MEDICA exhibition was the acquisition of Meditronics (Mumbai, India), a leading manufacturer of general-purpose X-ray products in India, by Royal Philips Electronics (Amsterdam, the Netherlands), the parent of Philips Healthcare (Best, the Netherlands). The acquisition of Meditronics comes just two months after Philips' acquisition of Alpha X-Ray Technologies, also based in India, and is indicative of the high-growth opportunities in emerging country markets as well as the attractive valuations for companies in the healthcare sector as a result of weakness in the financial market.
New players in POC testing
As an example of the growing number of companies involved in the point-of-care (POC) testing market, two major players in the patient monitoring market, Philips Healthcare and Dräger Medical (Lübeck, Germany), both exhibited new POC testing products in the sprawling MEDICA exhibition space.
Philips exhibited the Magnotech magnetic nanoparticle immunoassay system, a rapid, high-sensitivity IVD analyzer designed for use in mobile testing. The assay technology has been developed internally by Philips, and is based on the company's DVD technology. In addition to high sensitivity, the technology offers very rapid turnaround time, a must for POC applications. The system employs single-use microfluidic test cartridges and can perform both sandwich and competitive immunoassays, making it appropriate for both highly sensitive tests for hormones and proteins as well as for tests such as drugs-of-abuse assays.
For sandwich assays, antibodies on the magnetic nanoparticles bind to a target analyte, and second antibodies bound to the surface of the cartridge then bind to a second site on the analyte. By performing the second antibody binding step in the presence of an attractive magnetic field, the target molecules bound to the nanoparticles become highly concentrated at the binding surface, greatly decreasing reaction time.
Incubation times for immunoassays are typically accelerated 100-fold compared to letting the nanoparticles diffuse to the active surface. After binding to the surface is complete, a wash step is performed to remove unbound particles. Detection is performed optically by detecting the presence and extent of the nanoparticles which remain selectively bound to the surface of the cartridge via a sensitive optical technique called frustrated total internal reflection.
Prototype assays have been demonstrated for cardiac Troponin I at a sensitivity of 250 femtomolar, and for parathyroid hormone and morphine, the latter exemplifying a common drugs-of-abuse assay. The initial test menu will include a drugs-of-abuse panel with a turnaround time of one minute using a 15-microliter to 20-microliter saliva sample, which will be targeted for use in roadside testing.
Philips plans to obtain CLIA-waived status for the Magnotech system and assays, and expects to launch the product around the end of 2009.
Dräger Medical has entered the POC drugs-of-abuse testing market with a multi-drug testing system that provides results for up to six different drugs in 5 to 10 minutes using a saliva sample. The test menu includes opiates, benzodiazepines, cocaine, ecstasy, amphetamines, and methamphetamines. The device employs lateral flow test strip technology, and includes a reader to perform color analysis.
The product complements the Dräger Alcotest breathalyzer alcohol analyzer, and was introduced this past summer in Europe. The Dräger drugs-of-abuse testing system is not cleared for sale in the U.S.
Radiometer Medical (Copenhagen, Denmark) has launched a new analyzer, the AQT90 Flex POC, which significantly expands the company's POC test portfolio, adding a wide range of rapid immunoassays to its existing blood gas/electrolyte/chemistry test systems.
Radiometer has historically been the leader in blood gas/electrolyte test systems for use in satellite and near-patient testing in Europe, but has not until now participated in the POC immunoassay segment dominated by suppliers such as Inverness Medical Innovations/Biosite (Waltham, Massachusetts), Siemens' Dade Behring unit (Deerfield, Illinois), and Roche Diagnostics (Indianapolis).
The new Radiometer system includes a dedicated blood sampling device, the Monuvette, for collection of a whole blood sample, and uses a multi-test reagent cassette capable of performing a mix of up to 15 different tests, with a menu that includes Troponin I, CKMB, myoglobin and ß-hCG. The ß-hCG assay was introduced at the MEDICA exhibition.
Tests under development include D-dimer, Troponin T, NT-proBNP, CRP, and high-sensitivity CRP. Tests require a 100 microliter whole blood sample, and results are provided in 18 minutes using enzyme immunoassay technology. Radiometer plans to add additional coagulation tests to the menu this year.
Another new POC test system for cardiac marker analysis is under development by Micropoint Bioscience (Sunnyvale, California). The Micropoint mLabs Platform employs fluorescence immunoassay technology based on a lateral flow format. A unique feature of the system is its use of an electrowetting technology to control reagent flow through the test cartridge. The mLabs technology provides high precision (3% to 5% around the cut-off point), a 10-minute turnaround time, and a 100-microliter to 250-microliter sample volume requirement.
Tests under development include BNP, Troponin I, D-dimer, and tetanus. Micropoint is preparing to begin clinical trials to generate data for a regulatory approval submission in China, with plans to introduce the product in China in 3Q09. The company also plans to obtain regulatory clearance in the U.S. as well as obtaining a CE mark.
A new POC test system for hemoglobin A1c measurement was introduced at the MEDICA exhibition by Quotient Diagnostics (Surrey, UK). The Quo-Test A1C analyzer employs a boronate affinity assay technology that uses a combination measurement of fluorescence quenching and absorbance to determine the glycated hemoglobin percentage, and provides imprecision of less than 3% and a four minute turnaround time using a 4 microliter whole blood sample.
The Quo-Test is a new POC testing platform that will be expanded in the future to include other POC parameters. The Quo-Test A1C is now being sold in the UK, and Quotient is searching for distributors for additional country markets.
LifeAssays (Lund, Sweden) introduced a new POC test system for high-sensitivity CRP analysis at MEDICA, the LifeAssays hsCRP. hsCRP was recently found to be a powerful independent marker of cardiovascular risk in the JUPITER trial, which evaluated outcomes for patients treated with CRESTOR, a statin drug from AstraZeneca (London), and found a 47% risk reduction in patients with elevated hsCRP but normal LDL who were treated with a 2-fold higher statin dose.
The new LifeAssays hsCRP will enable rapid (six-minute) determinations of cardiovascular risk using a 20-microliter whole blood sample at the point of care. A normal range CRP test also is available for the LifeAssays system, and the company plans to add additional tests to the menu, including urine albumin and whole blood Cystatin C.
One major unmet need in point-of-care testing is for cardiac marker tests that can provide earlier detection of myocardial infarction and stroke. Existing markers such as Troponin I and T may not become positive until three hours or longer after an event, while myoglobin, although providing earlier detection than cardiac troponin, is not highly specific for MI.
rennesens (Berlin, Germany) has developed a new POC test that may enable earlier detection of a heart attack with improved specificity. The test detects heart fatty acid bind protein (hFABP), a marker that becomes positive as early as 20 minutes after an infarction, and provides a definitive indication after an additional 15 minutes. The rennesens CardioDetect is a rapid, lateral flow immunoassay test that provides results in 20 minutes using compact test cartridges.
MeDaVinci (Amsterdam, the Netherlands), in partnership with Demecal Europe (Haarlem, the Netherlands), is developing an even more rapid hFABP test for point-of-care use that combines a simple plasma separation device from Demecal with rapid qualitative immunoassay technology from MeDaVinci. A test result can be generated in only 90 seconds with visual readout, avoiding the need for a reader and the associated cost and complexity.
The company also is evaluating a brain-related FABP marker that may provide an early indication of ischemic stroke. MeDaVinci plans to introduce the hFABP test in 3Q09.
The hospital-based point-of-care testing market in Europe targeted by new POC testing products introduced at the MEDICA exhibition is projected to exhibit strong growth through 2012, as shown in Table 1. Growth is expected to outpace that for the overall clinical diagnostics market in Europe. Particularly strong growth is projected for the critical care (blood gas/electrolyte/chemistry), coagulation, and whole blood glucose segments.
 |
A new patient-attached point-of-care monitor for ex vivo measurement of blood analytes is under development by Sphere Medical (Cambridge, UK). The Sphere Proxima system consists of a compact bedside monitor and a disposable multiparameter blood microanalyzer that can be connected to an arterial line. Blood is drawn into the arterial line and over the sensor, which immediately takes a measurement, with the result displayed on the bedside monitor. The sensor chip can remain connected to the arterial line for up to three days, and measures a panel which can include pH, pCO2, pO2, glucose, lactate, potassium, sodium, ionized calcium, and hematocrit.
A second-generation sensor under development measures the intravenous anesthetic propofol. Additional analytes under consideration include antibiotics, anesthetics, sedatives and anticoagulants. Readings can be taken as frequently as every one to two minutes, enabling applications such as therapy titration for patients in the ICU. Other important benefits of the Proxima system include elimination of blood loss, since all blood is returned to the patient, and reduced infection risk since caregivers are not required to handle blood samples.
The system is being manufactured by Analogic (Peabody, Massachusetts), and is being evaluated in in vitro studies in preparation for starting clinical trials. Product launch is targeted for late 2009 or early 2010.
Outside-the-hospital monitoring products
New patient monitoring products were also described at MEDICA targeted at applications outside the hospital. Scientific Intake (Atlanta) exhibited the Sensor Monitored Alimentary Restriction Therapy (SMART) device, a non-invasive bariatric therapy system. The SMART device is designed to limit food intake by simulating Torus palatinus, a condition that physically restricts dietary intake at the mouth. The device forces patients to take smaller bites and chew their food thoroughly, limits bolus size and eliminates gulping, allow the patient to fully taste and savor food, and permits triggering of the satiety response.
An embedded microsensor detects when the device is inserted to monitor compliance, and a reader allows a record of device use to be uploaded via an infrared link to the patient's computer and thence via the Internet to a secure website. The patient's physician can log on to monitor progress of the weight reduction program.
A pivotal clinical trial has been completed which demonstrated a 5% weight loss in patients using the SMART device compared to a 0.4% weight gain in controls over four months. The company expected to receive a CE mark by year-end 2008, and plans to launch the product in Europe and Canada early this year, followed by U.S. launch after FDA clearance.
Scientific Intake said it believes there is a large potential market for the SMART device, with 134 million adults in the U.S., 135 million in Europe, and over 2 billion worldwide in the body mass index range of 27 to 35 indicated for treatment. That is more than 10 times larger than the population eligible for bariatric surgery.
Aipermon (Munich, Germany) exhibited the AiperSunny and AiperMotion 440 activity monitors, which are designed for use by patients undergoing weight control treatment. The AiperSunny is a €59 patient-worn device that tracks activity using a miniature accelerometer and signals when the patient's daily activity goal has been reached. The AiperMotion 440 is a more sophisticated monitor priced at €250 that not only monitors activity levels but that also enables the user to input data on caloric intake for computation of a caloric balance.
Another new non-invasive patient monitor was exhibited by ATES Medica Device (Verona, Italy) for ambulatory monitoring of EEG that could prove particularly useful for management of epilepsy patients, which number 50 million worldwide according to the U.S. National Institute for Neurological Disorders and Stroke. The ATES GEM system is used much like a Holter monitor, but for tracking EEG data rather than ECG.
ATES has established a partnership with Electrical Geodesics (Eugene, Oregon), which manufactures the dEEG dense-array EEG electrode, a scarf-like electrode array that allows reliable monitoring and ease of attachment. The ATES system, currently at the prototype stage, consists of a compact recorder connected to the EGI electrode allowing EEG signals to be recorded for up to two days, and can be used as a portable sleep laboratory as well as for monitoring epilepsy patients to detect and characterize seizures.
Non-invasive monitoring of blood analytes is becoming more widespread as a number of new devices have entered the market. NBR Optical Systems (Wuppertal, Germany) has developed a new device, the Haemospect, which provides real-time quantitative monitoring of hemoglobin levels without the need to draw blood. The device is CE-marked but not yet FDA-cleared.
It measures total hemoglobin, oxygenated hemoglobin and vasomotion, and computes oxygen saturation. Applications include non-invasive detection of intra-cranial hemorrhage, intra-operative bleeding, oxygen demand, and sepsis, the latter via analysis of changes in capillary filling time. The Haemospect is priced at $6,200.
Masimo (Irvine, California) also has introduced non-invasive hemoglobin as a new parameter for its Radical-7 and Rad-87 oxygen saturation monitors. The parameter can be used in transfusion management, to more effectively titrate blood and blood replacement products, as well as in detection of occult bleeding and real-time anemia detection. Masimo plans to introduce a spot-check device for non-invasive hemoglobin measurement this year.
Remote management of chronic disease
Non-invasive monitoring technology is also enabling a revolution in the management of chronic diseases such as diabetes, heart failure, asthma and respiratory disease, heart rhythm disorders, and hypertension. Diabetes is a major public health problem in Europe, as it is in the U.S. and worldwide. As shown in Table 2 on page 10, the number of diabetic patients in only five major European countries is similar to the U.S., and the number of amputations exceeds that for the U.S.
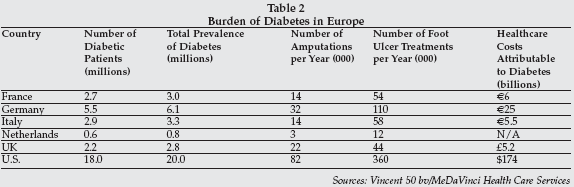 |
The burden of diabetes is expected to continue to increase not only in the developed countries but also in countries such as India and China, where the number of diabetics (40.9 million and 39.8 million respectively, according to the International Diabetes Federation) far outnumbers those in the western region. Worldwide, there are an estimated 246 million diabetics, and the number is projected to grow by 3% per year to over 380 million by 2025.
A number of companies introduced new products at MEDICA that are intended to improve diabetes management and help reduce the rate of diabetes-related complications. Vincent 50 bv, a unit of MeDaVinci, introduced a new remote monitoring service for detection of diabetic foot ulcers, one of the more common complications of the disease. The patented system includes the Vincent 50 foot scanner, which includes a camera to capture a visual image as well as sensors that detect oxygen saturation patterns and thermal images, enabling detection of developing ulcers at an early stage.
Pilot studies have shown that the Vincent 50 scanner has an 80% sensitivity for detection of foot ulcers as compared to visual examination. The images are uploaded to a call center and reviewed by an expert, compared with previous images to detect changes over time. Image analysis software has been developed for the call center that provides automated detection and data management.
The system is intended to enable ulcers to be detected earlier, particularly for patients with poor eyesight, obesity, stiff joints and neuropathy, factors that make it difficult for ulcers to be reliably detected at an early stage by patient self-inspection. The company has recently received a €2.1 million grant to fund development, and is in discussions with Philips Lifeline for marketing of the system in the U.S. Vincent bv is seeking additional partners for other regions.
Remote monitoring of blood glucose levels is another technology for improving the management of diabetes. BodyTel (Bad Wildungen, Germany) has introduced the GlucoTel Monitoring and Management System which includes a blood glucose meter with a built-in Bluetooth capability that links to the patient's cell phone to automatically transmit glucose readings to a call center. The meter requires a 0.6 microliter blood sample and generates results in 10 seconds. Real-time alerts can be generated at the call center to enable prompt treatment to help prevent complications.
The AllMedicus unit of H3 System (Daejeon, South Korea) introduced a new generation of its remote blood glucose monitoring system, the GlucoDr, a component of the GlucoAutoCheck Tele-Healthcare System. The system employs the recently launched DHS-5000 telehealth terminal from Intel (Santa Clara, California) to collect data from the GlucoDr glucometer and transmit the data to a call center.
The system can also monitor blood pressure, and in the future will have the capability to monitor body weight and heart rate. AllMedicus has partnered with Korea Telecom to establish a telemedicine network in Korea which now has more than 2,000 users.
TaiDoc Technology (Taipei, Taiwan) launched a new telehealth system at MEDICA that also will enable users to remotely monitor blood glucose as well as blood pressure. TaiDoc is a manufacturer of blood glucose and vital signs monitoring products with annual revenue projected to reach $50 million in 2008, up substantially vs. $30 million in 2007. The new telehealth system was developed in collaboration with leading hospitals in Taiwan.
Remote telemonitoring of vital signs is a relatively mature technology, and already is widely utilized worldwide for detection of conditions such as atrial fibrillation and other cardiac arrhythmias. However, advances in microelectronics technology are improving the ease and reliability of remote monitoring, and enabling a wider range of vital signs parameters to be tracked at low cost.
WPR Medical (Arendal, Norway) previewed the Curvus System, a wireless single-lead ambulatory ECG system that eliminates the connecting wires between the ECG lead and the monitor. A self-adhesive wireless ECG lead attaches to the patient's chest and connects to a handheld receiver via radiofrequency transmission. The receiver transmits the data via the GSM/GPRS cellular network to a monitoring center. Wireless network and Bluetooth interfaces are also available.
Use of the GSM/GPRS interface allows the patient to be monitored at virtually any location, and the wireless ECG lead enables monitoring while the patient is carrying out daily activities such as sports, gardening or taking a shower. The Curvus system is awaiting a CE mark, and is still in field testing, scheduled to be completed by year-end 2008. The single-use electrode will be priced at $200, and will allow monitoring for up to three days.
ECG is not transmitted continuously with the Curvus System. Instead, an algorithm detects arrhythmia events and data is only transmitted when an event is detected. A detailed ECG record can be downloaded when the patient visits the physician. The Curvus System is designed as a replacement for a Holter monitor, providing improved patient convenience and more rapid response to an event. The system is designed for use primarily in the hospital or in a hotel near a hospital, but also can be used in a nursing home, the patient's home, or essentially any other location.
VPATCH Medical Systems (Victoria, Australia) exhibited a similar wireless ECG monitor that may prove more cost-effective compared to the Curvus System. The VPATCH system provides 3-lead ECG monitoring using a disposable patch (the VPatch). A reusable microprocessor-based device, the V-POD, plugs into the patch and provides wireless two-way RF communication and also analyzes and stores data from the VPatch in real time.
Data is transmitted from the V-POD to the V-CELL, a handheld or in-pocket device that connects to the Internet via the cellular telephone network. Data is sent to a server-based software system, V-CENTRAL, which formats the data for viewing by a physician. The VPATCH sensor is priced at $20, and can be used for up to seven days.
Microimpluse (Stavanger, Norway) exhibited a development-stage non-invasive monitoring system for the hospital and home care market. The company's Vital Sign Monitoring (VSM) system is based on ultra-wideband technology, which enables non-invasive, non-contact, wireless sensing of physiological parameters such as heart rate, respiration, arrhythmias, cardiac output, and arterial stiffness. Non-invasive detection of hemo- and pneumothorax also is under development.
Ultra-wideband technology involves the emission of ultra-short microwave pulses with a duration of pico- to nanoseconds at periodicity of nano- to microseconds, and was first commercialized for applications in ground-penetrating radar. Microimpulse is now developing the technology for medical applications, where it may provide advantages in ease of use and cost.
The VSM system does not require the use of patient-attached electrodes or other sensors, but instead is positioned a few centimeters above the patient's chest, making it quick and simple to set up and particularly applicable to monitoring in settings such as nursing homes, physician's offices, and emergency response vehicles. In the hospital setting, the system can also provide bed occupancy information.
Production cost for the system is less than $500, and the lack of a requirement for disposable electrodes means that cost for the user will be lower than existing patient monitors.
Another unique non-invasive patient analyzer was exhibited by the Pneumosonics subsidiary of ElectroSonics Medical (Cleveland). Pneumosonics is developing the PneumoScan device, a handheld, non-invasive system for detection of pneumothorax. Pneumothorax is a condition that is typically caused by blunt trauma to the chest, and results in formation of an air pocket in the pleural space around the lungs, making breathing more difficult. In some cases, the condition can lead to lung collapse and possibly death.
The Pneumosonics device employs microimpulse radar technology developed at Lawrence Livermore National Laboratory (Livermore, California) and licensed by ElectroSonics. To perform a measurement, the device is placed at eight points on the chest, and a red or green light illuminates to indicate presence or absence of pneumothorax. A measurement takes 10 to 15 seconds to complete. The company plans to begin clinical trials in February, and is targeting product launch in early 2010 pending regulatory clearance.
Wound management, regenerative med
A number of new products for wound management, including products for preventing of scars in surgical wounds, were introduced at MEDICA.
Medtrade Products (Cheshire, UK) introduced the Celox granular haemostat, a new patented first aid product that rapidly controls bleeding in penetrating wounds. Celox has been used in Iraq and Afghanistan by the U.S. Special Forces for wounded troops, and also has been used successfully by police and fire departments in controlled testing.
Celox consists of a specially formulated biocompatible material which complexes red blood cells, forming a sealing plug in a wound that is then covered with a conventional compression bandage. The typical time to wound stasis using Celox is 30 seconds. After a wound has been sealed and normal clotting has occurred, the Celox material is removed with a saline wash.
A new version of the product, OMNI-STAT, is awaiting CE-mark approval, which is intended for use in cardiac surgery. OMNI-STAT will have an advantage compared to other types of wound sealants in that setting since it does not depend on the clotting cascade to achieve hemostasis. Consequently, it can be used to stop bleeding in patients who have received Coumadin or heparin, and also is effective in hypothermic conditions.
Ekkyo (Bouches-du-Rhone, France) exhibited the EkkyLite system, a laser-based skin healing and scar prevention device. The product has recently received a CE mark, and is being launched throughout Europe. The EkkyLite system is primarily intended for use following surgery, to promote healing of surgical incisions without scarring.
The technology is based on the use of laser light to generate highly localized heating of tissue to 50°C for 3 seconds, stimulating production of heat shock protein which in turn generates secretion of transforming growth factors beta 1 and 3 to promote accelerated healing and aesthetic tissue repair. Treatment of a typical surgical incision takes about 10 seconds.
The system consists of a handheld, battery-powered laser and a disposable Safety Strip. The Safety Strip contains a window through which the laser light penetrates to the skin, and a microchip embedded in the strip monitors the light dose. The laser unit communicates with the chip via a wireless interface to control the dose level, heating the tissue to the required temperature while avoiding burns. A back-up, non-contact infrared thermometer built into the laser unit is included as a safety measure.
The laser unit is priced at €15-20,000, and the disposable Safety Strips are priced at €8 each for a 1 cm strip. Cost for treating a typical 40 cm surgical incision is €500.
Another scar management product was exhibited by New Medical (Northbrook, Illinois). The company's NEWGEL+ is a medical-grade silicone product that is applied to scars to enhance healing and make scars less visible. It has been used successfully to flatten, soften and reduce discoloration of scars, and to prevent problem scars such as hypertrophic and Keloid scars. NEWGEL+ has received a CE mark, and is now on the market in Europe.
Next-generation therapies for wound management, and for treatment of a wide range of disorders ranging from neurologic diseases to orthopedic disorders, are employing tissue engineering and regenerative medicine technologies.
AxoGen (Alachua, Florida) exhibited the Avance Nerve Graft at the meeting, an implantable device for repair, replacement and reconstruction of damaged peripheral nerves. It is indicated primarily for repair of nerves damaged by trauma and as a result of surgery. Avance is a decellularized human allograft, treated to remove molecular constituents that cause immune rejection. The product is available in various lengths and diameters, and is processed and distributed in accordance with FDA regulations.
The graft is processed to preserve the 3-D structure of the nerve and the inherent growth simulating properties of the tissue, while clearing materials that inhibit axon regeneration. The graft provides a sterile scaffold that allows the natural healing process to repair peripheral nerves via tissue regeneration.
Another tissue-engineered product, but one intended for treatment of cartilage and bone defects, was exhibited by Sewon Cellontech (Seoul, South Korea). The company's Chondron product is designed to regenerate damaged or degraded cartilage in the knee.
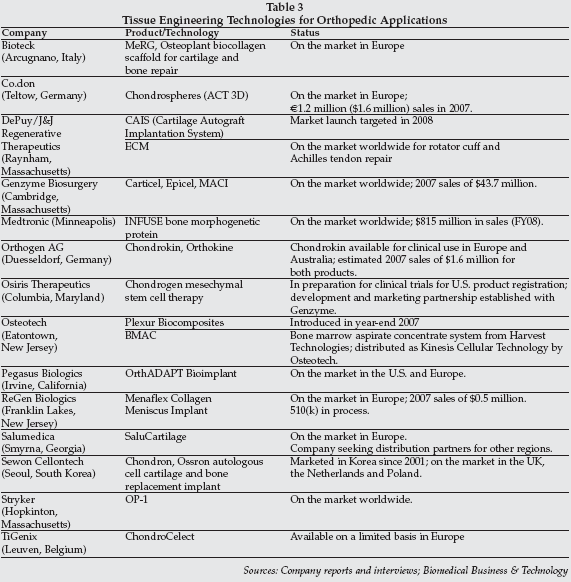
As shown in Table 3 on page 12, numerous companies have commercialized tissue-engineered products for cartilage and bone repair, including Sewon. Chondron is similar to Genzyme's (Cambridge, Massachusetts) market-leading Carticel, a product/service that involves harvesting of autologous chondrocytes, in vitro expansion of the cells at a separate laboratory facility, and subsequent re-implantation of the cells at a cartilage defect site.
 |
According to Sewon, Chondron has the advantages of ease of implantation, since no suturing is required; improved matching of the implant to the shape of the defect since it is applied via a syringe; and low cost. The implant procedure requires only 20 to 30 minutes, compared to three to four hours with competing products such as Carticel, and fibrin glue is used to hold the implant in place.
Chondron is now being marketed in South Korea and in Europe under a CE mark, but has not yet received FDA clearance. In South Korea, the total cost for a typical Chondron procedure is less than $10,000.
Sewon also has introduced Ossron, an implant designed for repair and replacement of bone.
Another cartilage regeneration product with applications in repair of knee and ankle cartilage defects, MeRG Biocollagen, was exhibited at MEDICA by Bioteck (Arcugnano, Italy). The Bioteck product does not require ex vivo culture of cells, but employs a Type 1 collagen scaffold which stimulates regeneration via mesenchymal stem cells. The process is based on the Steadman micro-fracture technique.
MeRG is an equine material, avoiding the infectious agent transmission concerns associated with bovine collagen, and, unlike products such as Chondron and Carticel that require an extended period of cell culture, can be taken from the shelf and used immediately, avoiding the need for a multi-step treatment process.
A new life-support system with applications outside the hospital, including uses in ambulances and helicopters, was introduced at MEDICA by Maquet (Rastatt, Germany). The company's CardioHelp is billed as the world's smallest heart-lung machine, and is designed for use in patients requiring temporary (14-day) heart-lung assist.
The device consists of a pump, an oxygenator, and integrated sensors for monitoring of hemoglobin, hematocrit, venous oxygen saturation, temperature and blood pressure. A single patient connection is required to establish extracorporeal circulation. The user interface is programmable so that emergency response personnel have a simple, easy-to-interpret interface appropriate for use in initial patient support and transport, while a more sophisticated interface can be provided for a perfusionist or a clinician in the ICU.
CardioHelp is designed to supplement ventilation and heart function, enabling the heart and lungs to heal following an acute event, and is sufficiently compact to allow it to be transported with the patient. The CardioHelp system will be available for sale in mid-2009.
