Articles Tagged with ''University of Pennsylvania''
Neurology/Psychiatric
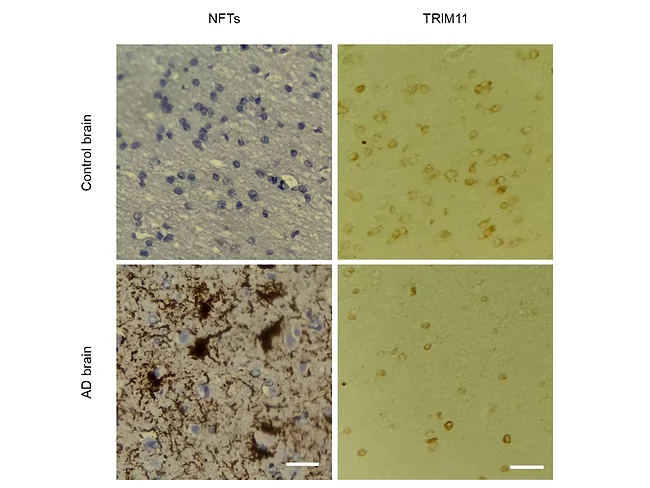
Quality control protein has multiple protective roles in tauopathies
Read MoreNeurology/Psychiatric
Identification and development of a PET radiotracer for imaging α-synucleinopathies
Read MoreDrug Design, Drug Delivery & Technologies